INSIGHTS ON REGULATORY COMPLIANCE
-
The Rising Cost Of Product Recalls In Life Sciences
Product recalls are becoming a major concern in life sciences. Learn why your company must act now to strengthen quality systems and reduce risk across pharmaceuticals, devices, and biologics.
-
What Pharmacies Often Miss In USP <800> Compliance
Master USP <800> compliance by transitioning from standalone equipment to an integrated system design. Review key protocols for a multi-layered strategy to mitigate cumulative drug risks.
-
Proactive EAM Automation For Compliance And Efficiency
Proactive compliance depends on automation. When asset data drives investigations, enforces change control, and reveals hidden failure patterns, compliance becomes an outcome of strong operations.
-
Avoiding Launch Delays With A Proactive Tech Transfer Strategy
Commercial success comes from treating tech transfer as a strategic collaboration. Flexible processes, early supply chain planning, and partnerships protect timelines and drive long‑term performance.
-
A Modular, Fully Automated Aseptic Processing Solution5/12/2026
Automated, no-touch aseptic processing enhances batch integrity and throughput. Explore how modular robotics and recipe-driven filling adapt to the complex demands of modern drug development.
-
Precision Medicine In IMIDs - The Path To Improved Healthcare Outcomes1/29/2026
Precision medicine is opening new possibilities for IMIDs by moving beyond symptom‑based care. Find out how emerging multi‑omics tools are reshaping how these complex diseases are understood.
-
Quality Assurance In Pharmaceutical Water For Injection (WFI) Systems3/19/2026
Maintaining the integrity of Water for Injection (WFI) systems is a non-negotiable priority in pharmaceutical manufacturing. Watch this webinar to refine your contamination control strategies.
-
Identification Of GLP-1 Analog Oligomeric States Using SEC-MALS7/29/2025
Discover how SEC-MALS enables precise characterization of GLP-1a therapeutics to ensure formulation integrity and enhance safety, efficacy, and regulatory confidence.
-
What Do You Need To Do To Qualify A Single-Use Assembly?8/28/2024
Learn about the implementation of single-use technologies, single-use user requirement specifications, and best practices for single-use qualification, including quality and regulatory documentation.
REGULATORY COMPLIANCE SOLUTIONS
-
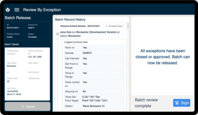
Enforce standardized workflows while collecting data in real time, giving you the visibility you need to prevent deviations, review exceptions, and release batches faster.
-
Peristaltic pump tubing can be a hidden source of variability and risk. See how advanced composite designs offer stronger durability, cleaner performance, and steadier flow.
-
The SecurityLINK fingertight fitting system simplifies your system and column connections and provides consistent performance with torque limiting technology that prevents column damaging overtightening.
-
Ensure product quality with a Hazard Analysis and Critical Control Points plan, with guidance from experts to keep your production process compliant and safe without sacrificing speed.
-
Streamline maintenance with a paperless platform that boosts uptime, ensures GMP compliance, and improves efficiency through automation, mobility, and KPI-driven insights.