Altering Patient Treatment: How Subcutaneous Delivery Can Help Patients Manage Chronic Conditions
By Victoria Morgan, West Pharmaceutical Services, Inc.

In recent years, the pharmaceutical industry has seen a steady shift toward increased patient centricity. The impact can be seen in nearly every aspect of the industry – from regulatory guidance, trial design, and drug delivery to new drugs proliferating the pipeline, such as biologics. Biologics are helping to revolutionize the treatment of chronic diseases such as multiple sclerosis and other autoimmune diseases, by helping patients take less frequent injections.
Additionally, by targeting specific components of a disease in ways never thought possible before, these therapies may also help some acute conditions, including certain types of cancer, become manageable chronic conditions.
A real focus of biologics research and development lies in new molecular entities which can be administered into the sub-cutaneous tissue in the skin. Therapeutic areas such as growth hormones and diabetes have long shown efficacy through sub-cutaneous delivery and an established patient acceptance of self-injection and pain tolerance. These therapeutic areas are rapidly being joined by oncology, autoimmune and blood disorders which traditionally had IV and infusion are the main routes of administration, but which are now seeing novel drug launches with sub-cutaneous delivery routes.

The sub-Cutaneous Space Is No Longer The New Frontier
Administration of large volume medicines has always been a challenge; one that has traditionally forced many drugs to be formulated into the <1mL space. This was the approximate volume which would be tolerated by the patient while still being an efficacious dose. Yet, there is a paradigm shift underway in terms of what is possible in a pain-tolerant larger volume injection; all because of new ways of accessing the sub-cutaneous space. Whereas intravenous infusions usually have to be administered in a hospital or in a doctor’s office, subcutaneous administration may be performed by a health care professional at the patient’s home or even by patient self-administration2. In addition, subcutaneous administration helps to treat patients with poor venous access or to spare patients' venous capital3. Subcutaneous administration is of particular benefit for long-term or chronic drug treatments. In addition, subcutaneous administration may be better tolerated as compared with intravenous administration, as the slow absorption may abrogate side effects related to high serum concentrations.
Let’s Talk Sub-Cutaneous
For patients diagnosed with haemophilia A, a typical treatment regime would be octolog alfa every 48 hours with numerous injections to treat on-demand bleeds. The total number of injections each week could easily be more than 10. Patients diagnosed with multiple sclerosis may relapse when adherence to therapeutic regimens wanes, forcing a 3-5 day hospital stay for IV steroids. While some diseases have moved the needle forward in terms of patient compliance, such as the regular use of insulin pens in diabetes, many people worldwide still suffer from the daily reminder and pain of their injections. The sub-cutaneous space allows us to think differently about how a patient perceives his or her illness by allowing larger volumes to be administered less frequently. When patients are not frequently reminded of their condition, adherence can be improved. Pharmaceutical companies recognise this, as do regulators, as is shown in Figure 2: the increasing number of sub-cutaneous approvals per year
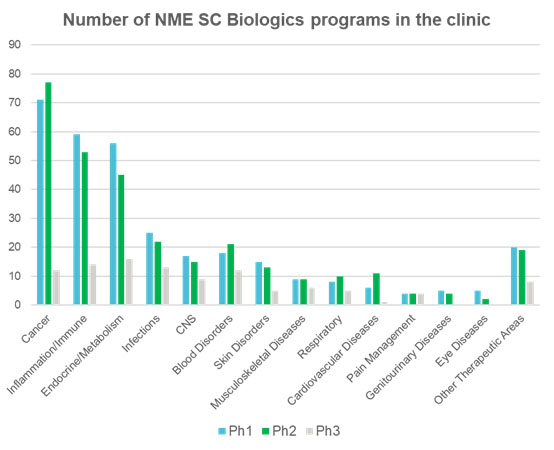
Benefits of sub-cutaneous delivery flows through to the clinic thanks to pharmacy efficiencies with faster drug prep time5, less set up reduces nursing time5 and fixed dosing reduces waste and medication error6. Sub-cutaneous administration is a win for the patient, the clinic and the pharmaceutical company while it offers a way of differentiating the product to gain (or in the case of biosimilar threat, slowing the loss of) market share.
Delivery devices as part of a combination product
Combining active pharmaceutical product with a novel sub-cutaneous delivery device makes joining the world of combination devices a well-timed one. Precedents have been set by Amgen with the launch of Onpro® for neutropenia during chemotherapy as well as Pushtronex® single use on-body infusor for hyperlipidemia. Amgen’s on-body infusor incorporates West Pharmaceutical Services, Inc.’s wearable drug delivery platform, the first generation of which was the first of its kind to be FDA approved in combination with an approved drug. These combination products have revolutionised the way a patient visualises their illness as they enable the patient to home administer their treatment thereby avoiding a repeat trip to the hospital or clinic.
There Is More To Come – From Device To Platform
In the drive to formulate biologics for patient adherence, higher volumes and higher viscosities are typical product profiles of many sub-cutaneous drugs. However, higher viscosities may not allow for conventional delivery due to the need for longer injection times to reduce patient discomfort. West recognised this trend and responded by developing its SmartDose® drug delivery platform, which includes a Gen. I device that allows up to 3.5mL of liquid drug to be administered over a longer period of time. First, human trials were conducted between 2011 and 2014 and development on wider-platform offerings – including large volume and preloaded options – started soon after. With Amgen’s FDA approval in 2016, EMA approval in 2017 and Japan/rest of world approval in 2018, global acceptance of combination products has begun. The SmartDose platform has expanded with a user loaded Gen. II up to 10mL device which leverages the success of the first-generation device with proven engineering and industrialization on a larger scale. With options available for dose volumes up to 10mL, the SmartDose Gen. II device can adapt to a variety of drug delivery needs.

Reassurance in Knowing SmartDose® Gen. II 10mL Device Is Patient Friendly
In the words of Alexander Pope in 1711, “to err is human7” and this is still very apt in 2019 when we think about human factors and user error. The simplest of devices in the hands of device engineers can become a behemoth in the hands of a patient; hence the importance of human factors in Phase III trials. Extensive human factor studies and user interface development has been done by West using the 10mL user loaded SmartDose® Gen. II device. This extensive work included Body Mass Index (BMI), age, previous injection experience as well as people who were patients (and took injections) and healthy volunteers (who did not take regular injections). The design usability, acceptability and comfort were assessed along with establishing whether the user needs were addressed and what the monthly dosing preference was. Results showed that the SmartDose Gen. II 10mL device had a well-received on-body size, was intuitive, easy to use and desirable. When treating a chronic condition with the need for an 8mL monthly dose, 7 total treatment options were evaluated. The SmartDose Gen. II 10mL device was voted the most preferable treatment compared to the other treatment options, which included weekly auto-injectors, monthly dosing via multiple, auto-injectors, visiting a clinic or via infusion. Patients don’t like taking frequent injections and a SmartDose Gen. II 10mL device monthly injection helped them with less frequent injections.
scPharmaceuticals and Alexion Choose West’s SmartDose® Drug Delivery System
In January 2019, scPharmaceuticals Inc. announced it had signed a development agreement with West Pharmaceutical Services, Inc. to incorporate its SmartDose® drug delivery system for delivery of FUROSCIX®, scPharmaceuticals’ lead program for the treatment of edema in patients with heart failure. scPharmaceuticals selected the SmartDose drug delivery system for its FUROSCIX treatment based on, in part, the features and functionality it offers for improving the overall patient experience.
During its recent Investor Day, Alexion announced their choice of the SmartDose® drug delivery platform for its clinical trial program for the delivery of ULTOMIRIS® once-weekly subcutaneous injections. West and Alexion have signed a development agreement for SmartDose® first generation and potentially for second generation development for exclusive use to delivery Alexion’s ULTOMIRIS® clinical development programme. ULTOMIRIS® is used in the treatment of Paroxysmal Nocturnal Hemoglobinuria (PNH), a chronic and debilitating, potentially life-threatening ultra-rare blood disorder. Using the SmartDose drug delivery platform, the drug has the potential to be the first to market subcutaneous option for PNH and atypical HUS (Hemolytic Uremic Syndrome).
SmartDose® Drug Delivery Platform Makes Development Easy
By partnering with West for an Integrated Solutions program that includes regulatory support and clinical filling of SmartDose® device cartridges, customers can ease their path to market for combination products. In 2019, West announced it has commenced discussions with Swissfillon AG, a provider of aseptic fill and finish services to pharmaceutical and biotechnology companies, that are intended to lead to a non-exclusive global collaboration to provide fill-finish capabilities to customers using the SmartDose platform for complex molecules.
Through the collaboration, it is anticipated that West will be able to deliver an integrated solution with filled Daikyo Crystal Zenith® cartridges for the SmartDose wearable device, which is expected to accelerate clinical development and enable customers to bring their innovative injectable drugs to market quickly. This new collaboration is expected to offer customers a robust fill-finish capability later this year.
Aiming for Success
Patients have never before been on the receiving end of such a rapid wave of advanced therapies, and biologics are at the heart of this wave. However, the ever-increasing list of demands a biologic drug places on drug developers is a significant challenge. In a price-sensitive, patient centric world, value has never been more needed. Working with a carefully selected partner, such as West, who has proof of performance and a collaborative spirit can be the difference in navigating the significant challenges found along the drug development road. Let us help you to improve your patients’ outcomes, avoid costly delays to launch and provide a better return on investment. By choosing the SmartDose® drug delivery system, you choose to you choose to improve the treatment experience of your patients as they confront their illness.
For example, insulin and some vaccines are given subcutaneously. The length of the needle used must be between 23 to 25 gauge and the volume of remedy administered is no larger than 1ml.
Subcutaneous injections are recommended for medicines that require slower absorption rates than in the case of intramuscular injections. By this method, substances that do not irritate the body tissues, are administered in adipose tissues.
For example, insulin and some vaccines are given subcutaneously. The length of the needle used must be between 23 to 25 gauge and the volume of remedy administered is no larger than 1ml.
Subcutaneous injections are recommended for medicines that require slower absorption rates than in the case of intramuscular injections. By this method, substances that do not irritate the body tissues, are administered in adipose tissues.
For example, insulin and some vaccines are given subcutaneously. The length of the needle used must be between 23 to 25 gauge and the volume of remedy administered is no larger than 1ml.
References
¹PharmaCirlce January 2019
2 Schweighofer CD and Wendtner CM (2010) First-line treatment of chronic lymphocytic leukemia: role of alemtuzumab. Onco Targets Ther 3:53–67
3 Launay-Vacher V (2013) An appraisal of subcutaneous trastuzumab: a new formulation meeting clinical needs. Cancer Chemother Pharmacol 72:1361–1367
5 Erwin De Cock et al. 2016, Cancer Medicine, p389
6 Jeroen JMA Hendrikx et al. 2017, The Oncologist, p1-11
7 Alexander Pope, An Essay on Criticism, Part II, 1711
SmartDose® is a registered trademark of West Pharma. Services IL, Ltd., a subsidiary of West Pharmaceutical Services, Inc., in the United States and other jurisdictions.
Crystal Zenith® is a registered trademark of Daikyo Seiko, Ltd. Daikyo Crystal Zenith® technology is licensed from Daikyo Seiko, Ltd.
Onpro® and Pushtronex® are registered trademarks of Amgen, Inc.
FUROSCIX® is a registered trademark of scPharmaceuticals, Inc.
ULTOMIRIS® is a registered trademark of Alexion Pharmaceuticals, Inc.
Important product and safety information for SelfDose™ Patient-Controlled Injector: https://www.westpharma.com/products/self-injection-platforms/selfdose
The SmartDose® on-body delivery system is intended for PRESENTATION/DEMONSTRATION PURPOSES ONLY. The device is not available for sale or use and will not be put into service until its conformity has been declared. Important product and safety information and warnings available at: https://www.westpharma.com/products/self-injection-platforms/smartdose/10-large-volume-wearable-injector-device

