Batch Quality Prediction In Pharmaceutical Manufacturing

Challenge
Quality is the most critical metric in pharmaceutical manufacturing – after all, nothing is more important than protecting patient health. Drug companies need to test each batch to ensure it meets quality standards.
But predicting the quality of a batch has traditionally been a challenge for drug manufacturers. The usual process is to take a sample while a process is running and send it to the lab for analysis. But waiting for lab results adds time – often several hours – to the process. And once the results come back, nothing more can be done to save a batch that was going bad. The quality is either acceptable or not. If the batch does not meet the quality requirements, the manufacturer will lose anywhere from hundreds of thousands to millions of dollars.
A large molecule pharmaceutical manufacturer was struggling to predict batch quality results in near real time. Delayed lab results made it difficult for the company to optimize process inputs to control the batch yield. The company’s process inputs were set with a known value, resulting in the potential of wasted energy and raw materials. The company needed a better way to predict batch quality, enabling process optimization.
Solution
Using Seeq, the scientists built a model of process quality based on data from the OSIsoft PI data historian. The team uses the model to predict the quality of future batches, enabling modifications during production before a batch needs to be scrapped for a quality issue.
This analysis uses reactor temperature, volume, and concentration as the critical process parameters for controlling yield. The raw data is filtered to the desired operation of interest, the reactor heating portion of the process. A predictive model for yield is then generated based on the maximum temperature, average volume, and maximum concentration. The model was deployed online to detect abnormal batches.
Results
Instead of waiting for quality tests to come back from the lab, the manufacturer has potentially saved millions of dollars by gaining the ability to rapidly identify and do root cause analysis of abnormal batches via modeling. It can reduce the number of out-of-specification batches by adjusting process parameters during the batch, a major breakthrough. The company also saved on the reduction of wasted energy and materials.
Developing and deploying an online predictive model of the product quality and yield can aid in fault detection and enable rapid root-cause analysis, helping to ensure quality standards are maintained with every batch.
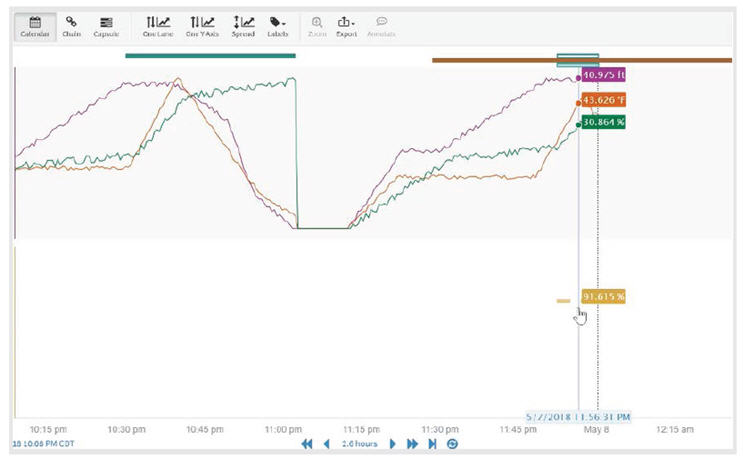
Batch quality prediction model compared to online data for the reactor heating portion of the batch.
