Cytiva Biopharma Manufacturing Solutions
Address biomanufacturing capacity with flexibility, speed and confidence.
Take control of your timelines
Adapt to evolving biomanufacturing demands and rapid capacity needs with modular facilities and integrated, flexible manufacturing platforms. Innovate and expand your production environment and workflow needs.
Flexible capacity transformed
Rapid and flexible capacity requires an optimized process design, adaptable manufacturing platforms, automation expansion capability and ongoing technical and mechanical support. See how biomanufacturers are leveraging our knowledge, experience and one point of contact management to ensure biomanufacturing resilience.
Discover flexible manufacturing options.
FlexFactory™ biomanufacturing train
Expand your biomanufacturing resilience to address future unknown needs with a flexible single-use platform. Your FlexFactory™ manufacturing platform is optimized for your needs today and readily adapts for scale-up or scale-out requirements. Gain efficiencies with automation options that can grow with your strategy.
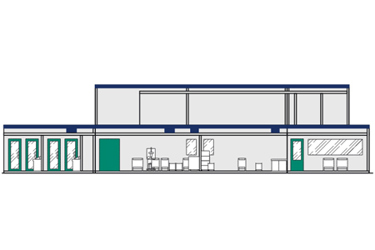
KUBio™ modular facilities
Comprehensive biomanufacturing solutions for cGMP production that dramatically reduce risk and time to market. A parallel production pathway enables reduced time to market and deferred CapEx investment until additional clinical trial data can be obtained. Your capacity expansion is streamlined with a suite of services and centralized project management.
KUBio™ box environment
KUBio™ box modular environments are designed to help manufacturers bring high-quality biopharmaceuticals to market more quickly. Available KUBio™ box modular environments for BSL-1 and BSL-2 can be delivered in 10 to 15 months for implementation inside existing infrastructure.
