From Pandemic Collaboration To Patent Litigation: BioNTech's Case Against Moderna
By Kevin E. Noonan, Ph.D., partner, McDonnell Boehnen Hulbert & Berghoff LLP

One of the paradoxically beneficial consequences of the COVID pandemic was that even without the compulsory aspects of the vaccine patent waiver proposed by certain World Trade Organization (WTO) member states (Brazil, India, and South Africa in particular), vaccine makers, especially Moderna and BioNTech/Pfizer, foreswore exercising their patent rights in favor of facilitating as widespread a distribution of vaccine as possible. This behavior was particularly significant considering the long-term investment in funding and scientific effort these technologies represented for these and other companies.
That was then.
On February 19, 2026, BioNTech filed a complaint in the U.S. District Court for the District of Delaware asserting that Moderna and its subsidiaries were infringing claims of BioNTech’s U.S. Patent No. 12,133,899, wherein claim 1 is representative:
1. A pharmaceutical composition comprising an RNA that:
(i) includes modified uridines in place of all uridines [which in dependent claims are each N1-methyl pseudouridine] and
(ii) comprises a nucleotide sequence that encodes a polypeptide, wherein the polypeptide comprises:
(a) one or more fragments of a SARS-CoV-2 spike (S) protein, wherein one of the fragments comprises a receptor binding domain (RBD),
(b) a secretory signal [which in dependent claims are from the SARS-CoV-2 S protein], and
(c) one or more additional domains selected from a trimerization domain [which in dependent claims are from T4 fibritin protein], a transmembrane domain [which in dependent claims are from the SARS-CoV-2 S protein], and a combination thereof;
wherein the RBD is linked to one of the additional domains via a linker, and wherein the linker comprises a GS [glycine-serine] linker.
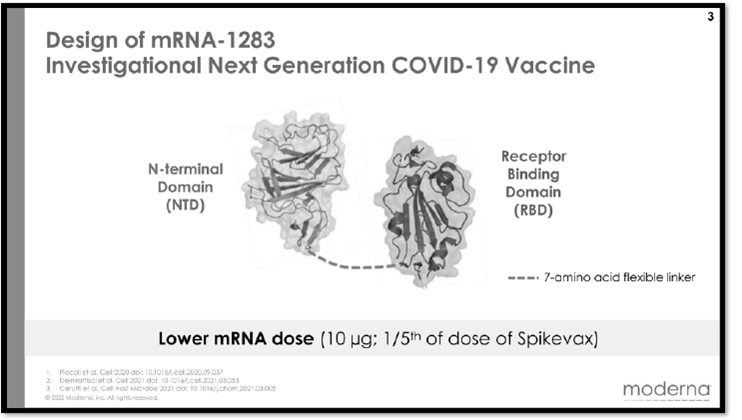
BioNTech accused Moderna of using its mRNA construct comprising these components in its mNEXSPIKE vaccine against COVID (SARS-CoV-2). According to the grievance, the claimed mRNA comprises a “streamlined vaccine design,” which triggers “an equally strong immune response to that of the complete spike protein” despite comprising only select domains thereof, specifically, a receptor binding domain, a secretory signal, a transmembrane domain, all from the SARS-CoV-s spike protein, and a trimerization domain from the heterologous T4 fibritin protein; the full-length spike protein structure is illustrated in the complaint as:
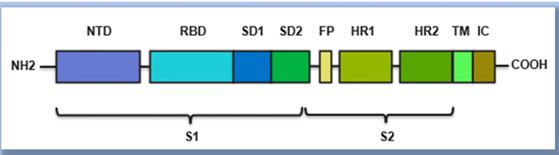
According to BioNTech the encoded protein mimics the full-length spike protein biology, as illustrated by these drawings:
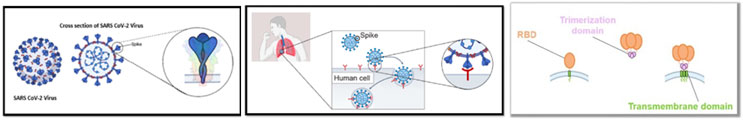
And the structure of the individual components and the “streamlined, domain-based spike” is shown here:
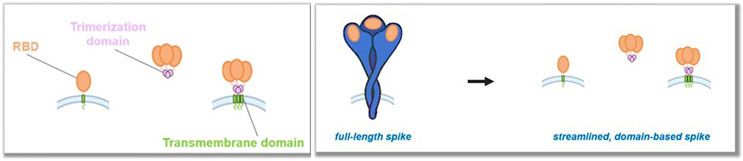
This vaccine can be given to patients at lower dosages and is more stable during storage and transport, according to the complaint. The complaint also highlights how Moderna itself lionizes its new (according to BioNTech, copied) form of their COVID vaccine, particularly regarding being able to administer a lower dose and the advantages of using lower dosages, with concomitant improvements in profitability.
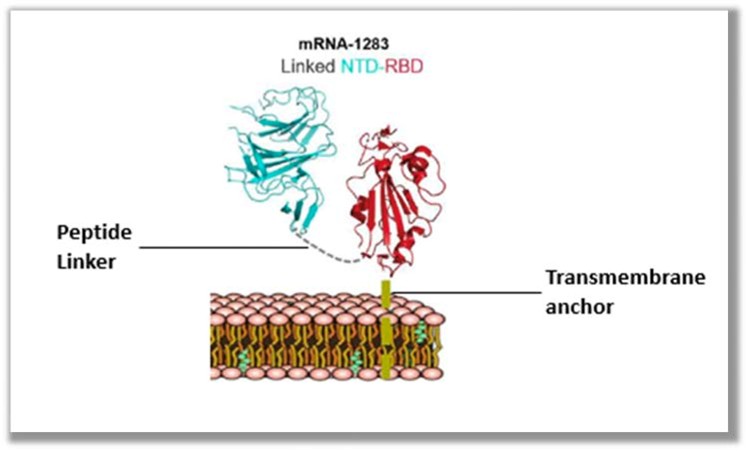
The basis for BioNTech’s infringement allegations is supported by the illustrations of Moderna’s mNEXSPIKE vaccine (below), accompanied by descriptions of the components in the label (including N1-methyl-pseudoruridine substitution and the presence of a secretory signal, a receptor binding domain, and a transmembrane domain joined with a peptide linker).
This is just the latest in a series of post-pandemic lawsuits brought by several companies (and patent holders) that have sued each other in the past few years. These lawsuits have involved either the lipid nanoparticle (LNP) component of COVID vaccines or the mRNA payload.
LNP technology has been the subject of extensive research and companies have been obtaining patent protection for several decades. Not surprisingly, patents for these technologies have been involved in several of the current lawsuits. Alnylam Pharmaceuticals v. Pfizer and Alnylam Pharmaceuticals Inc. v. Moderna Inc. resulted in settlements after several adverse decisions against Alnylam’s asserted claims in U.S. Patent Nos. 11,246,933 and 11,382,979; the parties agreed Moderna’s COVID vaccines did not infringe. Nevertheless, Alnylam has continued its lawsuit against BioNTech and Pfizer on similar claims.
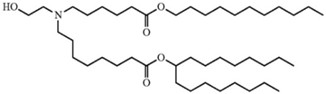
Arbutus sued Moderna on its claims of U.S. Patent 9,404,127 to “stable” nucleic acid lipid particles, which were invalidated in an inter partes review proceeding by the Patent Trial and Appeal Board (PTAB) and affirmed by the Federal Circuit. Those claims recited certain physicochemical structures (termed “non-lamellar”) but the Board held that these structures were inherently disclosed in earlier-filed applications. Arbutus has also sued BioNTech and Pfizer on claims to its LNP technology, as claimed in U.S. Patent Nos. 8,492,359; 9,504,651; 11,141,378; and 11,318,098 (see below).
On the other hand, Arbutus/Genevant and Moderna recently settled their lawsuit over LNP technology specifically comprising a particular cationic lipid claimed in U.S. Patent Nos. 8,058,069, 8,492,359, 8,822,668, 9,364,435, 9,504,651, and 11,141,378:
This settlement included an immediate (July 2026) payment by Moderna of $950 million with an additional $1.3 billion being subject to a determination on appeal regarding Moderna’s claim that these sums should be paid by the federal government because its COVID-19 vaccine, Spikemax, was produced under the provisions of 28 U.S.C. § 1498(a).
Moderna, for its part, also received a global nonexclusive license to patented technology for SM-102-containing mRNA vaccines for infectious diseases, specifically SPIKEVAX, mNEXSPIKE, and mRESVIA (according to Arbutus’s Form 8-K filing with the Securities and Exchange Commission on March 5, wherein the latter vaccine is directed to respiratory syncytial vaccine) and an agreement not to sue for “certain” (undisclosed) patents on Moderna’s products using the technology. It is unclear, however, whether Moderna’s vaccines against diseases other than infectious disease would fall within the scope of this license (e.g., Moderna’s anticancer vaccines, which have recently had positive clinical trial results).
Moderna has separately sued rivals Pfizer and BioNTech for infringing patents related to mRNA technology (U.S. Patents 10,702,600; 10,898,574; and 10,933,127, directed to beta-coronavirus mRNA). BioNTech challenged the ’600 and ’127 patents in inter partes review and the PTAB found them to be invalid; this decision is on appeal to the Federal Circuit.
BioNTech countersued Moderna in February, arguing Moderna’s next-generation COVID-19 shot, MNEXSPIKE, infringes its U.S. Patent No. 12,133,899.
A different and distinct patented component of this technology was the basis for Bayer to sue a number of COVID vaccine makers earlier this month. In those proceedings Bayer filed suit separately against Moderna, Inc.; Moderna U.S., Inc.; and ModernaTx Inc.; Pfizer Inc.; BioNTech SE; BioNTech Mfg. GmbH; and BioNTech U.S. Inc.; and Johnson & Johnson; Janssen Pharmaceutica NV; Janssen Pharmaceuticals Inc.; Janssen Biotech Inc.; and Janssen Vaccines & Prevention N.V. with respect to U.S. Patent No. 7,741,118. This technology is directed at the mRNA component of the vaccines, which reduces the number of polyadenylation signals (ATTTA) in an mRNA payload and as a result improves translation efficiency.
Finally, last summer BioNTech reached a settlement with Curevac over its U.S. Patent Nos. 10,760,070; 11,667,910; 11,786,590; 11,865,084; 11,883,486; 11,918,643; and 11,920,174 (among others, albeit not all of which were directed to coronavirus) over mRNA production and purification methods and pharmaceutical compositions. In a complex settlement, BioNTech acquired Curevac and GlaxoSmithKline received a monetary settlement in recompense for a failed joint venture with Curevac.
One significant feature of all these lawsuits: none of the plaintiffs has asked the court for an injunction, a remnant of the ethical positions taken by patentees during the pandemic.
Going forward, various combinations of companies that have had variable successes with developing and marketing COVID-19 vaccines are involved with or have concluded (by settlement or otherwise) their disputes over patents for technologies that were developed over time. The question now is the extent to which vaccines and other developing therapeutics will rely on these fundamental patents (and their relatives adapted to other uses, such as chronic diseases) and the extent to which, absent the pressures created by a global pandemic, the motivation/incentive for at least some level of cooperation remains for the benefit of mankind.
About The Expert:
 Kevin E. Noonan, Ph.D., is a partner at McDonnell Boehnen Hulbert & Berghoff LLP in Chicago and co-chair of its Biotechnology and Pharma Practice Group. Noonan is experienced in all aspects of patenting and in biotechnology and chemistry, representing pharmaceutical and biotechnology companies, as well as universities.
Kevin E. Noonan, Ph.D., is a partner at McDonnell Boehnen Hulbert & Berghoff LLP in Chicago and co-chair of its Biotechnology and Pharma Practice Group. Noonan is experienced in all aspects of patenting and in biotechnology and chemistry, representing pharmaceutical and biotechnology companies, as well as universities.
He is a founder of the biotechnology and pharma patent law and news weblog, Patent Docs (www.patentdocs.org). In addition to his blog, Noonan writes and lectures extensively on biotechnology patent law.
Kevin earned a Bacvhelor of Science from the State University of New York, a Ph.D. in molecular biology from Princeton University, and a J.D., cum laude, from the John Marshall Law School in Chicago.
