From Vision To Vial – Dalton Pharma Services
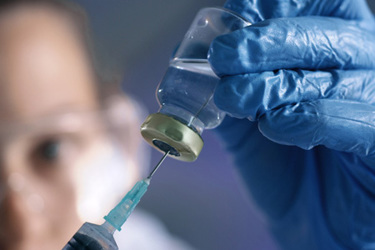
Bringing a molecule from discovery to market is rarely linear. It requires seamless coordination across development, manufacturing, quality, and regulatory functions - without disruption or delay.
Dalton Pharma Services is built around that integration. With more than 37 years of experience, our single‑site CDMO model brings API development and cGMP manufacturing, formulation, aseptic fill/finish, and full QC and microbiology testing together under one unified quality system.
By eliminating external technology transfer and reducing operational complexity, our model ensures continuity from early development through commercial supply. Backed by a globally experienced team and thousands of successful production campaigns, we help clients move programs forward with confidence and control.
Download the brochure for an overview of Dalton’s integrated capabilities and how we support programs end‑to‑end - from vision to vial.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Pharmaceutical Online? Subscribe today.
