Genentech's Heart Attack Treatment Approved by FDA
TNKase is a bioengineered plasminogen activator that is similar in activity to Activase (Alteplase, recombinant), a recombinant DNA-derived version of naturally-occurring tissue plasminogen activator (t-PA). But unlike Activase, TNKase has unique features specifically designed to prolong its half life, enabling single bolus dosing. It is also designed to increase specificity for fibrin, a key component of intracoronary clots and results in less disturbance of the body's natural clotting system.
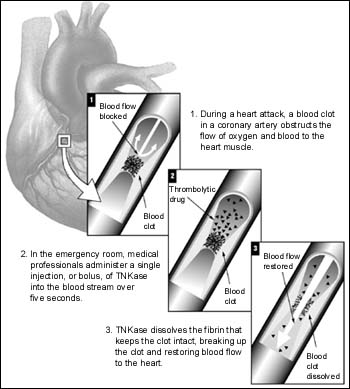
The FDA's approval of TNKase was based on Genentech's ASSENT 2 (ASsessment of the Safety and Efficacy of a New Thrombolytic agent) trial, a 17,000-patient study that compared a weight-adjusted dose of TNKase with the approved 90-minute accelerated infusion of Activase. TNKase was dosed in a weight-tiered fashion ranging from 30—50 mg based on weight.
According to the study results, TNKase and Activase were comparable in terms of mortality rates, yielding a rate of 6.2% (n=8461 and 8488, respectively). The treatments also displayed the same intracranial hemorrhage rate of 0.9%, and they had similar rates for stroke (Activase, 1.7 percent; TNKase, 1.8 percent). In addition, non-intracranial major bleeding and the need for blood transfusions were lower in patients treated with TNKase compared to Activase.
TNKase currently is under regulatory review by the European Regulatory Authority for the treatment of heart attack. Genentech's international marketing partner, Boehringer Ingelheim, will market the product in Europe, following approval.
Genentech Inc. is a biotechnology company that discovers, develops, manufactures, and markets human pharmaceuticals for significant unmet medical needs.
For more information: Betsy Rosenberg, Genentech Inc., 1 DNA Way, South San Francisco, CA 94080-4990. Tel: 650-225-7848.
