High Purity Polyethylene Liners For Pharma / Bio Applications: HiPure™ ULP7

A New PE Film for Bio-Pharma with Purity & Performance. HiPure™ ULP7 is a new pharma ready LLDPE film developed by ChargePoint Technology to meet the demands of even the most critical aseptic processing environments
HiPure™ ULP7 Film is a high purity polyethylene liner for use in pharmaceutical and biotech applications. The film is primarily used within ChargeBag® flexible packaging for the contained and sterile transfer of powder ingredients and as primary packaging for drug product transport between facilities.
The film is a single layer Polyethylene extrusion with excellent mechanical strength characteristics to withstand the rigours of industrial use giving operators and production teams a high level of protection from failures often associated with poor quality films.
The material is compliant to pharma standards and manufactured in controlled clean-room conditions to provide the basis for a consistent and ultimately sterile end product suitable for critical pharma application.
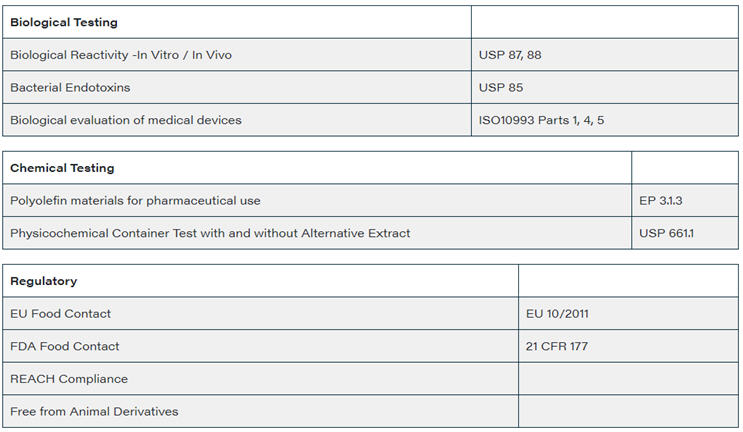
The Film has been tested to all the major regulatory standards and has achieved a well-rounded data profile for both chemical and biological tests that prove its suitability for a wide range of pharmaceutical products and uses minimal additives (purity) to ensure suitability for, and compatibility with, many pharmaceutical powder based products and production processes.
A permanent, non-migrating anti-static additive gives the film excellent surface resistivity properties to enhance performance with powders and qualifies as being Electrically Dissipative.
Regulatory Compliance – Testing
As part of the development of the film, ChargePoint have carried out rigorous and extensive testing of the HiPure&trade ULP7. The following qualification testing has been carried out and a pass rate has been confirmed: