8 mm Needle – Improving Subcutaneous Chronic Drug Delivery
By Aurelie Pager, Clinical & Human Factors Program Leader, BD Medical – Pharmaceutical Systems
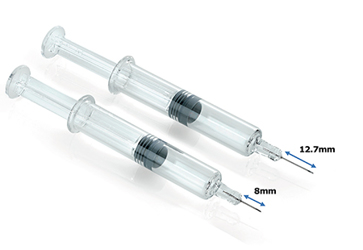
Chronic diseases are on the rise worldwide1,2 and require regular administration of drug therapies to relieve the patients from their symptoms. Biologics, and more specifically monoclonal antibodies (mAbs), are becoming dominant in the biopharmaceutical pipeline thanks to their success in treating chronic conditions, and biologics are expected to account for ~50% of top-100 drug sales by 2024.3 The parenteral route is commonly used, with >80% of chronic biologics delivered subcutaneously using prefilled syringes.4,5
Great progress and efforts have been made in offering drug delivery systems for chronic diseases over the past decades. However, several market unmet needs persist6 and should be considered when developing new manual drug delivery solutions. They include the need to:
- reduce the injection/needle-related anxiety and pain perception
- reduce the risk of accidental intramuscular (IM) injection
- maintain acceptable injection force and time for high-volume and high-viscosity drugs.
To improve patient quality of life and potentially treatment adherence, reducing injection frequency can be one of the strategies adopted by pharma companies. A way to achieve this could be to increase drug dosage7-9 either by increasing the concentration of the drug and/or by delivering a larger volume. Both options may lead to specific challenges.
Increasing the concentration of a biologic solution consequently increases its viscosity. Viscous and large-volume solutions take more force and/or require more time to perform the injection, which can impact end-user (healthcare providers, caregivers and self-injecting patients) acceptability.
Needle innovations, including the use of a shorter 8 mm needle, offer drugs, to enable the delivery of larger, more viscous drugs, without compromising end-user experience.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Pharmaceutical Online? Subscribe today.
