Modular, Single-Use Facilities Increase Biomanufacturing Flexibility

Introduction
The pipelines of biopharmaceutical companies are becoming increasingly diverse while improvements in cell lines are leading to more productive bioprocesses. These factors are driving new capacity demands for biopharmaceutical companies and their CDMO partners. It is becoming increasingly important, therefore, that companies implement Innovative, highly efficient, flexible facilities that are capable of meeting these challenging capacity demands.
New Biomanufacturing Facilities must meet the following requirements1
- Apply single-use technologies for lower capital expenditures (CAPEX), from cell vial to drug product vial and from development to routine manufacturing
- Provide a platform for rapid transition between product development and commercialization
- Ensure rapid start-up and ease of compliance – Be reusable and flexible as process and products change (New manufacturing strategies)
- Allow process transfers readily
- Allow the development of automation and control strategies, including PAT, data analysis, real-time release
- Allow security of supply (single-source vs. multiple sourcing)
Flexible Manufacturing with Single-Use Systems
Flexible manufacturing environments and single-use systems have eliminated many of the classical constraints on biopharmaceutical processes and have enabled designers to engineer modular bioprocesses and house them easily in properly classified environments. The ability to repurpose, improve, change, and reuse these core elements of a facility is having an impact on both large pharma and small biotech companies.
Modular, single-use technologies are providing companies with a clear alternative to fixed piped, stainless-steel equipment. Through effective partnerships with leading suppliers capable of providing innovative technologies, biopharmaceutical companies will be able to implement their future manufacturing strategies.
Optimized Layouts for Flexible Facilities
Sartorius has many examples of optimized facility layouts offering a considerably reduced footprint (Figure 1). Higher classification cleanroom areas can be considerably downsized by working with single-use closed systems. Large-volume support solutions can be stationed in a CNC (controlled non-classified) environment and fed through walls via transfer hatches and into higher classification areas.
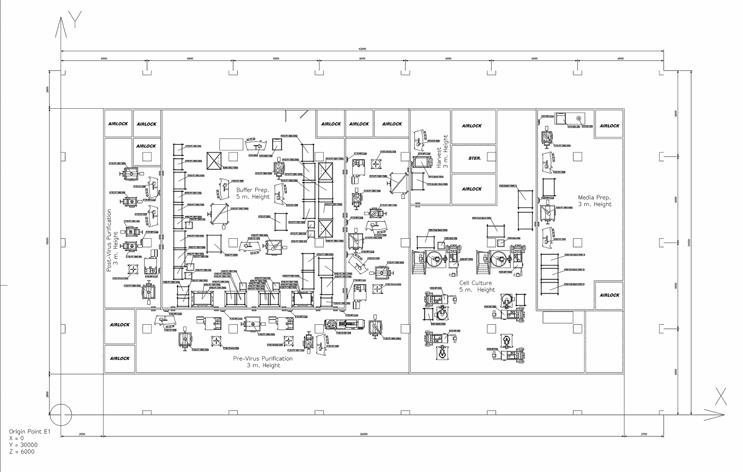
Figure 1: U-turn layout of a flexible mAb manufacturing facility
Modular Facility Concepts
Sartorius has developed modular facility concepts. Modular containers offer flexible modular systems that are perfect examples of how to enable the industry to cope with the challenges of the future. Figure 2 illustrates the application of such technology to a typical mAb facility equipped with Sartorius’ single-use process platform technologies.
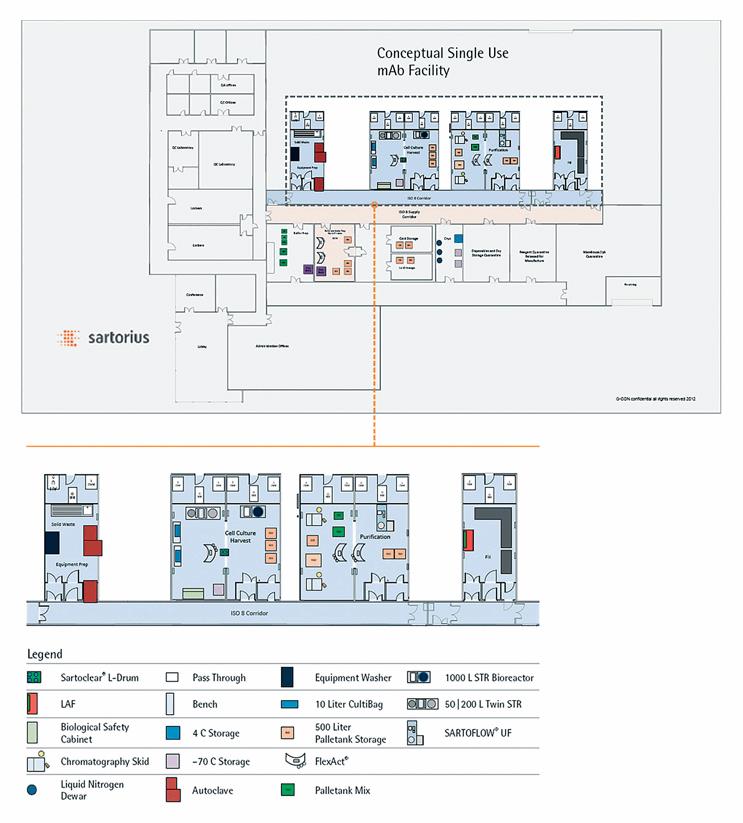
Figure 2: Modular facility equipped with Sartorius mAb platform process
Automation Engineering
Successful project execution requires not only consideration of the bioprocess equipment, but also its automation and integration into a given IT infrastructure. Yet there is no unique standard in the pharmaceutical industry, and many customers are using a variety of components (sensors, actuators, local controllers, DCS, SCADA, etc.) from different vendors. Once the user has clearly defined requirements, automation engineers are able to translate these into a functional specification and afterward into a hardware and software design specification followed by a design qualification phase.
In parallel, we offer our customers different automation architecture options. This can be based on either our proprietary solutions (DCU, MFCS) or on industrial solutions (Siemens, Emerson, Rockwell) or even, where appropriate, a mixture of the two. In this way, Sartorius offers full flexibility in automation and can offer the most suitable solution for our customer’s requirements. We are able to create and build the solution and carry out the respective testing accordingly.
Who are Sartorius Integrated Solutions?
Sartorius Integrated Solutions develops and implements rapid and cost-effective biomanufacturing solutions from early phase development through scale-up to commercial manufacturing.
1 Langer, E.S. 2016. 4 Key Trends in Biomanufacturing in 2016. (Based on a report from BioPlan Associates Bioprocess Online.)
