Performance Validation: HPAPI Containment Testing in A Risk-Based Era
By Michael Avraam , Global Product Manager

A surge in oncology and immune-suppressant therapies and increasing demand for high potency active pharmaceutical ingredients (HPAPIs) is pushing manufacturers to explore innovative containment strategies to meet handling requirements.
As these containment strategies evolve, so too does the role of containment verification. There is a clear need to understand the potential variations in testing and differing interpretations of results.
This article will discuss the key considerations linked to containment performance testing, data collection methods and the interpretation of results.
Industry landscape
It’s well documented that the biopharma market is expanding, thanks largely to the global demand and growth in the oncology market. By the end of 2024, the cancer segment is projected to reach close to US $100bn in value, expanding at a compound annual growth rate CAGR of 6.5%[i]. This has led to an increased need for the development of potent compounds and an increase in conventional drug manufacturing using HPAPIs. The HPAPI market stood at a valuation of US$2.64 bn in 2014 and because of these trends, it is projected to be worth US$25.11 bn by 2023. Between 2015 and 2023, the market is expected to expand at a CAGR of 8.3%.[ii]
[i] https://www.persistencemarketresearch.com/market-research/sterile-injectable-drugs-market.asp
[ii] https://www.transparencymarketresearch.com/high-potency-active-pharmaceutical-ingredients-market.html
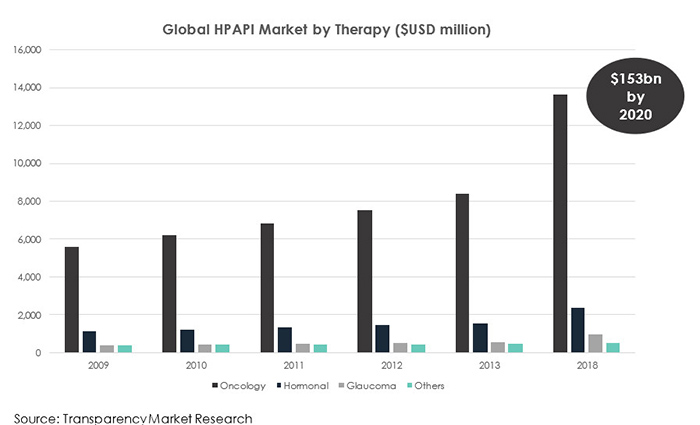
The containment solution market is expected to grow rapidly by 2020, resulting in an increasing need for more advanced control strategies in high potency manufacturing that address both the quality of the final product and, critically, operator safety.
Market diversification has allowed more innovative technology development to safeguard drug products and the operator. Isolators, restricted access barrier systems (RABS) and split butterfly valves (SBVs), are all now commonly used throughout the manufacturing process. Closed transfer valves, such as SBVs, are increasingly replacing traditional open transfer valves due to the limited manual intervention involved, which reduces the risk of cross contamination and the presence of airborne dust particulate, ultimately protecting operator safety.
Containment performance testing
All manufacturing equipment must be validated ahead of its use in the manufacturing process. For example, before a manufacturer can implement a new control device within its process, it should be assessed in line with the International Society for Pharmaceutical Engineering’s (ISPE) SMEPAC (Standardized Measurement of Equipment Particulate Airborne Concentration) guideline for its particulate containment performance. This is intended only as a guide for manufactures to enable them to demonstrate how a containment device will perform as part of a laboratory condition test, not within a process in the final manufacturing facility.
This guide was introduced in the 1990s following the focus from occupational health professionals on worker exposure measurement as the primary target to qualify containment equipment. It was formalised as SMEPAC and later adopted and revised by the ISPE. While it is widely welcomed by the industry today to ensure good practice within the validation of containment performance, the random nature of the guidance on sampling methods and distribution has been questioned by some who feel that it is challenging to achieve a specific measure of containment for equipment or devices. Some argue that the data lacks statistical validity and more importantly, the method would be better suited if it provided a baseline dataset for future integrity testing[i].
[i] http://www.pharmtech.com/environmental-containment-performance-there-accountability
Data interpretation
There are many factors that can affect the interpretation of the SMEPAC test results:
Placebos
The SMEPAC guide recommends a variety of particle sizes and levels of detection of placebos during validation testing, including lactose, paracetamol, mannitol and naproxen. However, many are sceptical of the results that come from SMEPAC tests. For example, many question how relevant it is to test placebos as they are so different to the real-life APIs that will eventually be used. In addition to this, many suppliers do not use the same placebos, so the results can vary considerably.
Test equipment
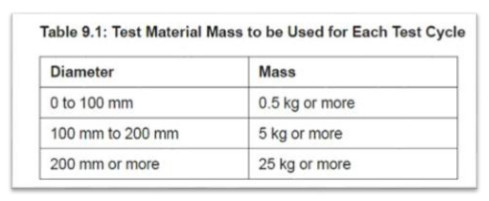
There is the possibility for test equipment with the same performance to show differing results due to the considerable differences from various samplers when using the same test. Figure 2 is an example test table demonstrating the test material mass range used for each test cycle.
Testing protocols
As detailed by SMEPAC, the testing protocols allow for a certain amount of inconsistency. For example, referring to transfer quantity, the SMEPAC guideline notes that ‘the masses are intended to fully coat the exposed seal and operating surfaces and are suggestions’ so the very nature of providing a suggested weight range means that testing devices with volume variation will likely result in inconsistent results.
Data
Data is an integral part of any test and manufacturers often use the data from this laboratory validation test to qualify the selection of the required containment technology for their process. However, this could prove to be misrepresentative, especially as the comparison isn’t like for like. There are variations in the way the containment performance tests are carried out in the laboratory and the interpretation and utilisation of the results obtained can be inconsistent with the real environment. Consequently, it is risky to presume that performance should be the same, and it’s important to consider the potential variables.
Contamination & operator intervention
During high potency manufacturing, it’s essential to ensure full operator safety and reduced levels of contamination. As human intervention is present at almost every stage of pharmaceutical manufacturing processes, solutions must counter the potential risks. Critically, containment needs to be achieved while not hindering productivity and operability, which can sometimes be challenging.
Containment validation testing must also reflect operator intervention to ensure that the containment device is tested accordingly. However, some of which can be reliant on operator technique to achieve performance thereby further increasing variability.
Potential exposure
Validation must also be carried out at each step where potential exposure is present in its normal environment, including a full risk assessment for the whole process. For example, a charging application that has not undergone contained dispensing operation prior to being within the laboratory environment, cannot be compared measurably to its normal application within the manufacturing area.
Preventative maintenance
Ensuring frequent monitoring and preventative maintenance helps to safeguard the reliability of the containment solution. Risk is limited if the identification and rectification of any damage has occurred prior to the test.
By limiting manual intervention throughout the whole validation testing process, this will also help to maintain a more consistent result.
Technological outlook
The containment market has continuously adopted new design technologies, including SBVs, which have evolved over the last two decades to address the more stringent containment demands when handling potent compounds. Other containment solutions such as isolators can be integrated with SBVs to enable the safer transfer of potent compounds. They can be used in many applications when, not only dust control and containment is a concern, but where product flow, yield and sterility are also important.
There are multiple ways to improve valve performance, including double gloving, enhanced wiping procedures and waste disposal. However, by further reducing human intervention and introducing an automated approach, could this be the answer to improved performance?
As the industry moves toward automation, the adoption of wireless monitoring technology will make it possible to receive vital equipment performance data and generate an audit trail much easier and quicker, thereby allowing maintenance, health and safety and compliance teams to make informed decisions to proactively manage their maintenance and validation programs.
Such innovative technologies promise to revolutionize traditional containment strategies allowing manufactures to meet the most stringent regulations. There is also a real potential for an automated approach to enhance performance validation monitoring by provider a real-time, ‘real-world’ method of validating and confirming equipment performance.
Final thought
While widely adopted and welcomed, there are key considerations when containment testing in line with the SMEPAC guidelines. It’s important for manufactures to understand the clear differences between laboratory and manufacturing environments and the potential limitations of these guidelines. With the introduction of a more automated approach to validation testing, there is the opportunity to capture more repeatable and reliable data, thereby improving the levels of containment performance. Perhaps the laboratory test could even be replaced in the future?
References
[1] https://www.persistencemarketresearch.com/market-research/sterile-injectable-drugs-market.asp
[3] http://www.pharmtech.com/environmental-containment-performance-there-accountability
