Pharmacoeconomics Beneficial to Both Pharmaceutical Companies and Patients

Datamonitor's new report, Optimizing Pharmacoeconomic Return on Investment: Benefits throughout the product lifecycle, reveals that pharmacoeconomics:
- is a powerful marketing tool for targeting healthcare payors, and
- can increase patient access to cost-effective drugs.
Targeting Helathcare Payors
Over the last year, several European governments have introduced pharmacoeconomic guidelines to curb healthcare costs and to optimize patient access to cost-effective treatments. Markets with such guidelines now include the UK, The Netherlands, Finland, Portugal, Norway, and Italy. Since it is likely that more and more markets will follow suit, pharmaceutical companies will be required to show the pharmacoeconomic value of their products on a regular basis.
If pharmaceutical companies can prove the cost-effectiveness of their new products, these results can be used in marketing efforts to a wide range of audiences. Datamonitor found in its primary research that the most important target audiences for pharmacoeconomic data are physicians and payors, as illustrated in the following diagram. Their importance is related to their key roles in prescribing and reimbursement decisions. In fact, pharmacoeconomic data are a powerful marketing tool in all markets, irrespective of guidelines, since they show payors the value and cost of treatment regimens within a restricted budget, often including indirect costs and benefits of specific treatments.

Pharmacoeconomics Can Increase Patient Access To Cost-Effective Drugs
Patient groups have met some of the recent pharmacoeconomic regulations with concern, since they fear that access to high cost products might be restricted. For example, a recent UK cost utility study including 132 patients showed that the treatment of secondary progressive multiple sclerosis (MS) with beta interferons is not cost-effective (Forbes, et al., 1999). Therefore, the study concluded that the money could be spent more effectively on other means of improving MS patients' quality of life.
However, there are opportunities for beta interferons to be judged cost-effective in patients with relapsing/remitting MS. The UK National Institute of Clinical Excellence (NICE) will issue extensive guidelines for these drugs by August 2000. NICE is taking into account overall costs of MS treatment, such as home care and social security costs. Therefore, if NICE deems the drugs to be cost-effective for relapsing/remitting MS, both pharmaceutical companies and patients will benefit from broader access to beta interferons.
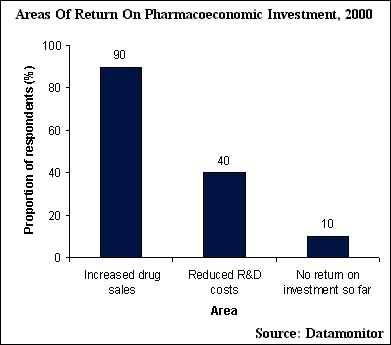
Overall, pharmacoeconomics has the potential to increase patient access to high quality healthcare and pharmaceutical drug sales. In Datamonitor's primary research with pharmaceutical executives, 90% of respondents believe that increased drug sales are a major area of return on pharmacoeconomic investment, as illustrated in the following diagram. The rising drug sales will be driven by greater coverage of so far untapped patient populations and, therefore, by increased patient access to high quality healthcare.
Optimizing Pharmacoeconomic Return on Investment: Benefits throughout the product lifecycle is available from Datamonitor, priced at $3,995
References
- Forbes, R.B et al. (1999). Population based cost utility study of interferon beta-1b in secondary progressive multiple sclerosis. British Medical Journal 319: 1529-1533.
For more information: Elisabeth Overend-Freeman, Datamonitor, 1 Park Ave., New York, NY. Tel: 212-686-7400, ext. 765. Fax: 212-686-2626. Email: efreeman@datamonitor.com
