Rethinking Product Development Through Lipid-Based Formulations
By Eduardo Jule, Ph.D., principal consultant, evolve Consulting
It is now common knowledge that the vast majority of new chemical entities emerging out of discovery display limited water solubility, resulting in poor and variable oral bioavailability. As a result, development of suitable drug delivery strategies has become a reality of everyday pharmaceutical development. To this end, formulation scientists have access to an ever-expanding toolbox: from straightforward processes such as particle size reduction to technologies requiring advanced (e.g., lipid-based) formulations (LBF) or process engineering expertise (e.g., amorphous solid dispersions [ASD] though spray drying or hot melt extrusion).1
Choosing The Correct Technology For Development
Scientists, often managing portfolios of drug candidates and under time pressure, may be prey to trial-and-error, blanket parallel testing, or “the technology (or vendor) you know” pitfalls. Trial-and-error simply evaluates against a series of platforms of increasing complexity, whereas parallel thinking looks at multiple techniques at once, in both cases to see what works best. While there is merit to either — collecting data and valuable experience with the active pharmaceutical ingredients (API) while limiting expenditure — end choices cannot always be properly and scientifically justified and often result in excessive drug usage at a stage where supplies are precious.
The third path is usually that of least resistance, as it deflects the choice to either a known technique or a vendor with whom we have had a positive prior experience. To be clear, if and when this third route is data- and experience-driven, then that is the right path. Oftentimes, though, drugs are force-fed into suboptimal formulations by innovators under constraints or by CDMOs looking to secure a deal in the one platform they offer or excel at, to some extent in a biased way. Technology selection matters, as it can have a substantial impact on product performance.
Why Optimization Matters
In 2011, Janssen’s Zytiga (abiraterone acetate [AA], 250 mg tablets) was approved by the FDA for the treatment of metastatic, castration-resistant prostate cancer in combination with prednisone or prednisolone, quickly becoming a standard of care. At the time of launch, the product displayed a label advising to “Take Zytiga on an empty stomach. Do not eat anything for at least 2 hours before or 1 hour after you take your dose.” While food effects are not uncommon, Cmax and AUC values were reported to jump up to 17x and 10x in the presence of food (high-fat meal), which poses a risk of highly variable — and thus unpredictable — blood levels and may result in side effects.2
In 2018, Sun Pharma’s Yonsa (AA, 125 tablets) was approved in combination with methyl-prednisolone at a lower dose, i.e., 500 mg vs. 1,000 mg for Zytiga. Such dose was achieved through the SoluMatrix Fine Particle Technology, a form of dry milling that reduces particle sizes from 3 µm to10 µm to 200 nm to 800 nm. This technique substantially increases the surface area, dissolution rate, and, ultimately, absorption, of AA particles. While this may seem a marginal achievement from a drug properties perspective, Yonsa demonstrated no food restrictions, increased absorption, and similar bioavailability to the originator,3 not only reducing doses by half but giving patients the flexibility to take treatment with or without food.4
From that perspective, relatively minor optimization efforts (particle size reduction) lead to major product performance enhancement that translates into actual patient benefits (increased bioavailability and reduced dosing, no food restrictions).
Understanding The Logic Behind Reformulation
Technology selection should be an informed, data-driven process supported by technical expertise. A careful literature analysis of the physical, chemical, and biological properties unveils AA is:
- poorly soluble (1 µg/mL)
- moderately crystalline (melting point ca. 150 degrees C)
- highly lipophilic (logP > 5).
These characteristics hint more to a grease ball than a brick dust, opening the possibility to formulate in an ASD, but most likely in an LBF. Noting the drug is subject to foods effects, our efforts focused on the latter, even if additional studies looked at an ASD and at multi-particulate forms for comparative purposes (data not shown).
First, a comprehensive solubility and stability screening of excipient chemical families led to the identification of a battery of options that were successively tested as single, binary, and, ultimately, ternary compositions. Composed of lipids, a surfactant, and a co-surfactant, these formulations were tested against key performance criteria, dispersion and digestion, using the in vitro protocols defined by the Lipid Formulation Classification System Consortium.5 AA concentrations in resulting phases (non-digested lipid, digested aqueous, and precipitated pellet) were evaluated, some over time, to further understand the ability of a given composition to solubilize and retain the drug in solution post dispersion and digestion.
Ideal formulations result in the highest AA concentrations in aqueous phases, from which it is more readily and consistently absorbed. The drug is dissolved in small dispersed colloids, including micelles and small vesicles, of mixed exogenous and endogenous materials. Screened through this systematic strategy (as well as against stability and machinability), lead formulations were encapsulated in two-piece hard capsules (liquid filled hard capsules [LFHCs]). Then, they were dosed against a single Zytiga tablet as a benchmark in crossover studies using beagles (n=6).
Tablets resulted in low and variable AA absorption, which was increased tenfold in the fed state, not dissimilar from what is observed in humans. LFHC, bearing our lead formulation, resulted in a fasted state exposure slightly higher than that of the tablet, only at a dose of 50 mg, i.e., 20% that of the originator. In addition, variability was substantially reduced, while absorption was nearly independent of the presence of food (figure 1).6
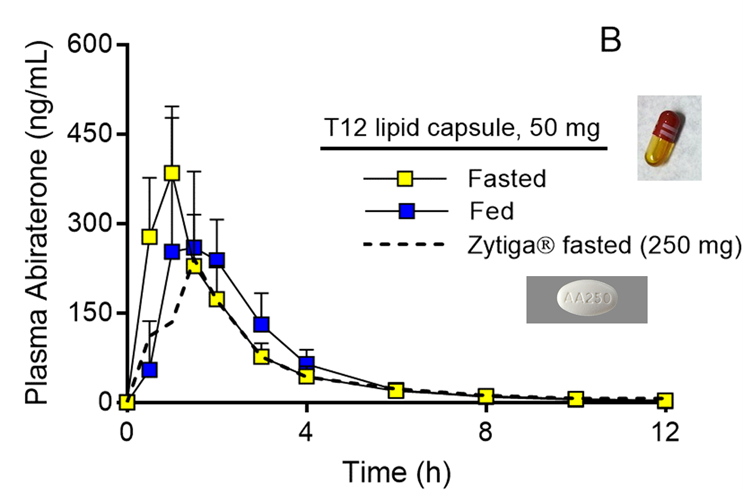
Figure 1: Abiraterone plasma concentration vs. time after oral administration of Zytiga tablet (250 mg AA) or LFHC (50 mg AA) to beagles in either fasted or fed states.6 Mean±SD
Formulation of AA, with a technology better suited to its physical, chemical, and biological properties, and thus more likely to effectively tackle its problem statement, led to:
- bioavailability enhancement
- concomitant dose reduction
- reduction of variability
- reduction of food effects.
Hypothetically (the product concept was never tested in clinical environments), this approach could lead to even further reduced dosing, less variable and more predicable blood levels, and eliminate food restrictions, all tangible benefits for patients. In the meantime, a new 500 mg strength of Zytiga was launched in 2017, partly superseding the potential benefits introduced here (pill burden, but not the ability to take with or without food).7
Technology Selection Matters: The Substantial Impact On Product Performance
From an innovator’s perspective, having the ability to more effectively evaluate safety and effectiveness, achieve proof-of-concept, and maximize return on investment through optimized drug utilization are all sound strategies for individual and portfolio management in early stages, when drug supplies are limited. As the product matures and reaches late-stage development, designing a next-generation target product profile (TPP) is also a sound strategy for individual and portfolio life cycle management. TPPs have the distinct added value of leveraging existing assets that are subject to lower attrition rates, if not commercially available.
Should the technical expertise not be available in-house, unbiased expert consultants may assist in generating TPPs, matching problem statements to enabling technologies based on available data, and designing pre- and formulation protocols. Unbiased experts can identify CDMOs to develop, evaluate, and manufacture formulation concepts that can be quickly supplied into animal, first-in-human, clinical, and commercial stages. After all, economy of materials, accelerated development pathways, and streamlined portfolio management combined with better clinical outcomes and tangible patient benefits are what pharmaceutical development is all about.
References:
- Williams, et al., Pharmacol Rev 2013, 65:315–499
- European Medicines Agency Product Information
- Goldwater et al., Clin Pharmacokinet. 2017, 56(7):803-813
- https://www.yonsarx.com/
- https://www.americanpharmaceuticalreview.com/Featured-Articles/154717-The-LFCS-Consortium-Supporting-Rational-Design-and-Testing-of-Lipid-Based-Formulations/
- US Patent 20200316093A1
- https://www.jnjlabels.com/package-insert/product-monograph/prescribing-information/ZYTIGA-pi.pdf
About The Author:
 Eduardo Jule, Ph.D., is a drug delivery scientist and formulation expert with more than 20 years of experience advancing complex problem statements from early development through clinical stages in CDMO environments. His background spans contributions to global pharmaceutical science initiatives, including the Lipid Formulation Classification System (LFCS) Consortium, leadership of product development teams, the ideation of bioavailability-enhancing formulations, and the generation of valuable intellectual property. As the founder of evolve Consulting, Jule advises biotechnology companies on target product profiling, technology selection, and outsourcing strategy; helping teams define their North Star, identify the right enabling platforms, de-risk decisions on partnering, and accelerate development timelines. He brings a unique blend of scientific depth, practical development insight, and execution discipline. He is a regular contributor to webinars, scientific conferences, and expert panels.
Eduardo Jule, Ph.D., is a drug delivery scientist and formulation expert with more than 20 years of experience advancing complex problem statements from early development through clinical stages in CDMO environments. His background spans contributions to global pharmaceutical science initiatives, including the Lipid Formulation Classification System (LFCS) Consortium, leadership of product development teams, the ideation of bioavailability-enhancing formulations, and the generation of valuable intellectual property. As the founder of evolve Consulting, Jule advises biotechnology companies on target product profiling, technology selection, and outsourcing strategy; helping teams define their North Star, identify the right enabling platforms, de-risk decisions on partnering, and accelerate development timelines. He brings a unique blend of scientific depth, practical development insight, and execution discipline. He is a regular contributor to webinars, scientific conferences, and expert panels.
