The Challenges Of Developing Combination Products: Regulatory Complexity And The Case For A Holistic Development Strategy
By Dr. Dave Seaward, Founder & Programmes Director, 3P innovation
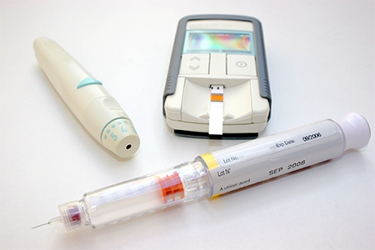
Combination products like prefilled syringes, autoinjectors, and inhalers now dominate pharmaceutical portfolios, yet their development complexity remains widely underestimated. Regulatory pathways diverge sharply between the US and EU, creating classification challenges that ripple through entire programs. More fundamentally, siloed approaches to formulation, device engineering, and manufacturing consistently trigger costly delays and rework.
Discover how early cross-functional alignment transforms outcomes, particularly when teams treat these projects as inherently complex rather than merely complicated. In this article, you'll learn why deferring equipment strategy until late-stage development creates a regulatory catch-22: clinical data requires representative products, but representative products need commercial-grade equipment. Proof-of-principle test rigs emerge as critical tools for de-risking device functionality, process parameters, and stability strategies before design freeze. The use of non-representative clinical supply for pivotal trials often invalidates stability data, forcing expensive repeat studies that could derail timelines.
Companies that integrate equipment planning, regulatory strategy, and representative manufacturing early don't just reduce risk—they fundamentally change program economics and speed to market.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Pharmaceutical Online? Subscribe today.
