
PRODUCTION

Solving The Analytics Bottleneck In Cell Line Development
Discover a fully automated, benchtop, walk-away system for accelerated and efficient cell culture sample preparation. Learn how this system clarifies and purifies mAbs in one step without sacrificing quality.

How To Select Your DSP Chromatography Technology?
Making the switch from single batch to multi-column chromatography can dramatically reduce resin utilization, costs and eliminate bottlenecks without sacrificing quality. This animated feature compares the traditional, single batch approach to a multi-column alternative f...

Slurry Bin Functionality With The DAC Ergo
Check out how our optional stainless-steel bin works in conjunction with the DAC Ergo to capture spent media.

Your Contract Manufacturer Partner
Discover an integrated solution for pharmaceutical companies that offers a broad range of manufacturing services and capabilities to produce high-quality devices.

How To Maintain A Nuclease Free Environment During mRNA Production
mRNA are singled-stranded, and their chemistry makes them more susceptible to enzyme degradation than double-stranded DNA. Learn how to maintain a nuclease free environment during mRNA production and purification.

Key Functions Of The ÄKTA Oligosynt Synthesizer
Discover a compact, fully automated oligonucleotide synthesizer for robust, scalable process development. Learn how it can help establish a robust and reproducible oligo synthesis process.

Improve The Bioavailability Of Poorly Soluble Drugs
KinetiSol is a fusion-based, solvent-free process that utilizes frictional and shear energies — in a fraction of the time of other amorphous solid dispersion technologies.

Scaling Up A Revolutionary Amorphous Solid Dispersion Platform
Process parameter and quality attribute relationships, including those for typical pre- and post-ASD unit operations, will be explored in the context of real-world examples.

Strategies And Emerging Technologies For mAb Capture
Explore opportunities with the introduction of high-capacity protein A resins, how and when multicolumn chromatography is beneficial, and how emerging technologies can address bottlenecks.
Meet The Experts: PAT, Raman Spectroscopy, And Chemometrics
Gain a better understanding of process analytical technology (PAT), Raman spectroscopy, chemometrics, and Raman applications in bioprocessing.

A Conceptual Overview Of The BioSMB MCC Platform
In this video, our technology expert Jason Forte describes how the BioSMB system offers 3-5 fold increase in productivity, yielding up to 80% savings on chromatography resin costs with minimal disruption to the accepted ways of doing chromatography today.

Singapore Single-Use Site Capacity Expansion
Thermo Fisher Scientific is expanding our single-use manufacturing network globally, an expansion site closer to Asia Pacific region

Transform Your Chromatography Processes With An Innovative Approach
Learn how to achieve better resin utilization, minimize disruption to existing methods, and align your strategy with unique process goals.

Client Sustainability Directives — CDMO Perspectives
Gain a deeper understanding of current directives and metrics to equip your team with the knowledge necessary to navigate the evolving landscape of sustainability.

Redefine Your Oligonucleotide Manufacturing
Streamline oligo production and scale confidently. Explore high-performing, purpose-built automated equipment designed to ensure safety, efficiency, and purity, from synthesis through concentration.

Thermo Fisher Scientific Suzhou, China Manufacturing Site Capabilities
Watch this video to learn more about this manufacturing site capabilities, quality management systems, PPI implementation, product expansion, assurance of supply and lead times.
How To Select Your DSP Chromatography Technology
Watch to explore the differences between the conventional single-column batch method and the multi-column approach for mAb capture.
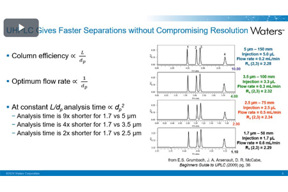
UPLC Columns: Fundamentals, Chemistries, And Selected Small-Molecule Applications
Discover the fundamentals of UPLC columns and their diverse chemistries. Learn more about versatility in separating small-molecule analytes using reversed-phase and hydrophilic interaction techniques.

Thermo Fisher Scientific Single-Use Global Manufacturing Network
We have implemented a multi-tier strategy to improve the resiliency of our supply chain and created the largest networked single-use manufacturing organization in the world.

The Fusion-Based, Solvent-Free Process Disrupting Spray Drying
Review a commercially-ready process capable of reproducing spray-dried dispersions with less manufacturing complexity and environmental impact, and out-designing and outperforming spray drying.

Ensure Speed And Success With Your Drug Delivery Project
Hear from experienced professionals in the drug delivery space on how to ensure success and speed-to-market by establishing a firm foundation when working with a CMO.

Small-Scale Oligonucleotide Synthesizer That Supports Scale Up
Learn about a small oligonucleotide synthesizer with big impact that features a user-friendly interface, compact footprint, and more. This small scale system is user friendly and supports scale up.
How To Plan For Trouble-Free Filter Validation
Watch to learn when filter validation should be performed, what is required from an end user and laboratory testing perspective, and how to achieve first-time success for various drug products.

Showcasing A Scalable Downstream Platform For Multiple AAV Serotypes
Watch to explore how to maximize AAV full capsid recovery, establish a scalable platform for various serotypes, and utilize analytical tools to measure empty/full capsid ratios and AAV titers.

Improve The Performance Of Your Small And Large Molecule Medicines
Explore the latest in nanotechnology advancements including recent clinical data, formulation applications, and API optimization to NanoImprove formulations.

Rapid Process Development And Technical Support For AAV Scaleup
Accelerate your AAV production journey. Learn how rapid process development and expert support can streamline your path from vial to purified bulk, ensuring scalable and efficient manufacturing.

Meet The Experts: Part 1 - Process Analytical Technology (PAT)
Watch part 1 of this 4-part interview series to learn about process analytical technology (PAT) from expert Stacy Shollenberger.

The Future Of mRNA Manufacturing
Watch to explore a box solution that provides flexible and scalable options for your mRNA workflows and is designed to support the evolving landscape of mRNA production.

Technology To Overcome Absorption Hurdles And Solubility Limitations
For poorly soluble drugs, overcoming absorption hurdles can hinder development. Explore a solution that empowers researchers to overcome solubility limitations and accelerate drug development.

The Route To Faster Microbial Quality Control
Join product and application experts Anne-Grit Klees, Carine Krebs, and Esther Welterlin to discover the benefits of using a new rapid microbial detection platform.
