
QUALITY ASSURANCE

Overcome The Hidden Complexities Of mRNA Process Development
Explore the challenges behind the perceived simplicity of mRNA process development, as well as characterization strategies and models for predicting process outcomes at early stages.
Meet The Experts: PAT, Raman Spectroscopy, And Chemometrics
Gain a better understanding of process analytical technology (PAT), Raman spectroscopy, chemometrics, and Raman applications in bioprocessing.

Revolutionizing Contract Manufacturing With A Unified QMS And MES Platform
Watch to gain expert insights on utilizing a closed-loop, single-platform approach and explore practical examples and data showcasing the advantages of integrating your QMS and MES.
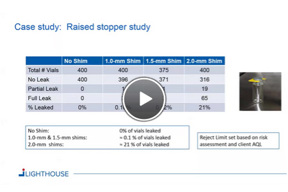
CCI Testing And Revisions To EU Annex 1
The new language in EU Annex 1 will likely have a significant impact on your CCI testing practices. In this webinar CCI testing strategies and the proposed revisions to EU Annex 1 are discussed.

A Rapid Approach For Moisture Determination Of Lyophilized Product
Explore the limitations of traditional moisture determination techniques, an innovative approach using laser-based headspace analysis, and real-world case studies using this non-destructive method.

Compressed Gas Risk Assessment: A Significant Step In Your CCS
Discover the importance of compressed gas monitoring in ensuring product quality and compliance with EU GMP standards.

The Pathway To Operational Readiness
Ready to ensure your facilities, systems, and teams are prepared for success from Day One? Discover a proven framework to accelerate operational readiness, mitigate risks, and achieve sustainable results.

Solutions In Pre-Filled Syringes For Biologics And Ophthalmic Applications
Discover how pharma companies can select tailored pre-filled syringes for biologics and biosimilars by aligning drug delivery needs with specialized containers and analytical services.

Autoinjector Testing: Addressing Sensitivity And Headspace Challenges
Micron‑level defects can jeopardize autoinjector sterility. Learn how vacuum decay offers a deterministic, repeatable way to verify container integrity and strengthen quality assurance.

Comprehensive CCI Solutions: From Testing To Consulting
Discover comprehensive CCI solutions that help ensure compliance, reliability, and confidence in your processes. Strengthen your quality strategy with expert-driven services designed for life sciences.

The Sensitivity Trap: Building Confidence In CCIT Performance
Effective CCIT goes beyond micron claims, focusing instead on repeatability, confidence, and real‑world relevance. Learn how to define a defensible LOD and understand what auditors truly prioritize.

Multi‑Sample Testing With Precise Failure Detection For Throughput
Test multiple samples at once with a multi‑sample testing platform that ensures accurate detection for low headspace formats and integrates into automated lines for high-volume efficiency.
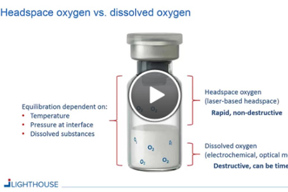
Determining And Controlling Oxygen Levels In Sensitive Formulations
This webinar will review how oxygen levels in finished parenteral drug containers can be determined and controlled throughout the product life cycle by using laser-based headspace analysis.

Ensuring Integrity: A Comprehensive Approach To CCI
Deterministic CCI testing ensures compliance in packaging. Learn how advanced methods like Vacuum Decay, HVLD, and Helium Leak Detection deliver results aligned with USP 1207 and global standards.

Precision And Reliability For Medical Device Packaging
Learn how to detect critical defects, reduce risk, and ensure compliance through flexible service options and data-driven testing that eliminates subjectivity in medical device packaging quality assurance.

Airflow Visualization Studies: The Impact Of Annex 1 On Sterility Assurance
Airflow visualization techniques like Smoke Studies and CFD analyses are crucial for contamination control, aiding cleanroom qualification, environmental monitoring, and optimizing contamination control strategies under Annex 1.

Minimizing Particulate Risk
While visible particles often draw immediate attention, visual inspection alone is insufficient, as subvisible particulate matter can pose equally significant risks to patient safety.

Navigating GxP Compliance Challenges
Learn how to select and manage trusted software suppliers to streamline your digital transformation, mitigate risks, and ensure regulatory adherence. Watch to discover best practices and real-world examples.

Meet The Experts: Part 1 - Process Analytical Technology (PAT)
Watch part 1 of this 4-part interview series to learn about process analytical technology (PAT) from expert Stacy Shollenberger.

Container Closure Integrity Testing Technologies - PTI Packaging Technologies & Inspection
PTI-Packaging Technologies & Inspection is headquartered in Hawthorne, New York, a Westchester county community close to New York City. We are a team of engineers, scientists and professionals serving the packaging industry as a leading equipment manufacturer of packa...

Using Positive Controls In Container Closure Integrity Studies
This webinar describes the use of positive controls as an important element of CCI studies designed to validate packaging components for CCI or to qualify processes for producing good CCI.
