Container Closure Integrity Testing: The Importance Of Sensitivity, Automation And Efficiency
By Oliver Stauffer and Michelle Wolf

Developments in non-destructive, deterministic testing technologies that can achieve optimum assurance in container closure integrity.
When assuring the quality of all packaged products, testing container closure integrity (CCI) is critical, especially as related to parenteral pharmaceuticals. If there is a breach of the seal, even a microscopic one, it affects the sterility and the stability of the product – the drug may not be as effective due to oxidation or moisture ingress impacting the active ingredient.
But more importantly, a defect in the seal can lead to patient safety concerns. Parenterals are sterile preparations intended to be administered directly into the systemic circulation in humans or animals (think solutions, suspensions, and powders for injection, infusion, or implants).1 A compromised parenteral container can act as a vehicle for microbial transmission and pose a significant risk to patient safety. Once bacteria find their way into the container, the drug is no longer a treatment but instead is a threat to the patient.
Therefore, packaging and closure of such containers must be up to high quality standards, as described by pharmacopeias. For example, the United States Pharmacopeia (USP) chapter 1207 “Package Integrity Evaluation—Sterile Products” provides guidance that emphasizes the importance of using reliable and sensitive methods for CCI testing and sets out recommendations on developing and validating a compliant CCI test.2
This article will explore the need for sensitive and fast automated CCI testing technologies that are also reliable and how existing solutions can help achieve optimum quality goals for pharmaceutical manufacturers.
Moving toward more reliable testing
Traditionally, to ensure that the container closure is preserving the sterility of a drug package throughout its shelf life, methods such as water bath, dye, and microbial ingress tests have been used. Particularly, dye ingress (also known as the liquid tracer test) is still widely used across the pharmaceutical industry due to its simplicity and versatility.3 However, these traditional methods are destructive, time-consuming, and limited in their effectiveness and reliability, as they are probabilistic methods. Often, probabilistic methods lack standardization within the industry. In addition, test operators may not be in control of certain test elements, and results obtained may be subjective.4
In light of these inadequacies, USP chapter 1207 was revised in 2016 to urge the industry to move away from probabilistic test methods in favor of deterministic ones that allow more control of variables, perform uniformly under different conditions, and detect even the smallest leaks with reliability.2
“There is a huge shift in the industry toward deterministic and quantitative test methods,” says Oliver Stauffer, Chief Executive Officer at PTI - Packaging Technologies & Inspection. “This includes vacuum decay, headspace analysis, and high voltage leakage detection (HVLD) for parenteral products. The industry is currently moving away from dye ingress and microbial ingress methods because there are so many blind spots in applying them and there’s a huge false sense of assurance.”
Vacuum decay is one deterministic method that is known to provide reproducible and reliable results for CCI testing of high-risk package applications. The test involves placing a package in a test chamber, applying vacuum, and monitoring the vacuum level for any change, which would indicate the presence of a leak.5
On the other hand, high voltage can also be used to quantitatively detect leaks without destroying the sample. When it is applied to a non-conductive package material that contains a semi conductive product inside, a spike in the voltage reading would indicate a defect in the packaging.6
Both methods have proven to be reliable non-destructive testing technologies and, depending on the specific characteristics of the product and container, one or the other can be chosen for inspection.
Testing challenges in the pharma industry
According to Stauffer, there are two main ways CCI testing is being approached in the industry today:
- 100% in-line testing: All products in a batch are tested at an acceptable level of CCI assurance in a production line at high speed
- Small batch automated testing: Products are tested in an optimal batch size that focuses on sensitivity and reliability, but inspection does not occur at the same rate as the packaging line
Most pharmaceutical companies adopt a 100% in-line inspection system for CCI testing, due to the speed and throughput that can be achieved. But it may not be the method that has the best standard of assurance. Many process-related defects can be missed when testing only at very high production speeds.
“I think it's really important to note that inspection speed doesn’t necessarily have to match the speed a manufacturing line is operating at – let’s say 500 products a minute – to make the whole operation efficient and cost-effective,” Stauffer says. “If you try to do 100% inspection in-line at that same rate, there is a huge gap in quality assurance and performance, because you are making speed your priority. When dealing with parenterals in a high-risk space, sensitivity and reliability should be priorities because the product can have a serious impact on the patient. Speed is something you can manage downstream.”

A majority of the 100% inspection systems are only able to detect leaks down to 20-25 µm,7 but in the parenteral space (especially for liquid-filled containers), defects below 10 µm can be critical. This is where small batch testing provides a benefit: there’s more flexibility to tailor the testing method to be extremely sensitive and detect tiny leaks that are just a few microns in size. Now, with the advent of robotics and other automation tools, small batch testing technologies are also gaining more speed.
“Regardless of the percentage of products in a batch you are testing, you should be testing with the highest confidence level to ensure results are appropriate,” Stauffer notes further. “The pharmaceutical industry operates with a very high standard of allowing only one in 10,000 false positives. With 100% in-line testing, you probably aren't going to be operating at that level of assurance for the level of integrity that you need.”
Focusing on 100% in-line inspection to save costs and ramp up production may result in a very cursory and inadequate approach to quality control of parenterals. According to Stauffer, most systems available on the market for 100% in-line testing don't have the level of sensitivity that is needed for these high-risk container formats.
How sensitivity, reliability, and speed connect in automated small batch testing
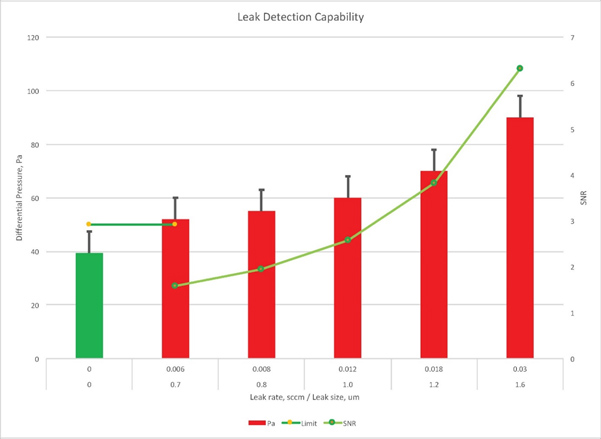
Sensitivity and reliability go hand in hand in CCI testing. When you increase the sensitivity of the test method, the overall measurement quality and reliability of that method may decrease. On the other hand, a more reliable inspection method may detect a certain size (or greater) defect but miss much smaller ones.
Finding a good balance between the two was a natural challenge for the industry until new technologies like vacuum decay and HVLD were optimized to allow both higher sensitivity and reliability, with added automation to rapidly test in small batches. “Now both these proven technologies have moved into the automated inspection realm, and all of a sudden, manufacturers have both a reliable test method that detects single-digit micron defects, as well as one that is inspecting at a speed on par with production operations,” claims Stauffer.
Automation would enable the inspection of about 25 to 50 samples per minute, with a heavy focus on the reliability and sensitivity of the signal. This allows a production line to also proceed with the highest level of quality assurance. As Stauffer puts it, “then you are running CCI tests at a pace that allows for the highest level of reliability and sensitivity, but in an automated fashion – so it's still maintaining the sort of integrated, automated approach that each production line requires.”
Such an automated platform would provide the same 100% testing capability with an accurate inspection that includes quantitative test data and a pass/fail result. Proper sensory measurement requires time to capture a measure. However, these new automated technologies can still reliably inspect containers at a much higher rate than was possible before, with more sensitive leak detection capability. Stauffer says this is something “the industry is shifting towards. If you are going to provide the quality and safety required for patient needs, then the time to make a good reliable inspection is absolutely needed.”
Developments in vacuum decay and high voltage leak detection
Vacuum decay is a renowned test method because it is non-invasive and non-subjective and can be applied to a wide variety of package formats and product types. The standard vacuum decay leak test method (ASTM F2338), developed using PTI's VeriPac instruments, is recognized by the FDA as a consensus standard for CCI testing and is referenced in the USP Chapter 1207.8
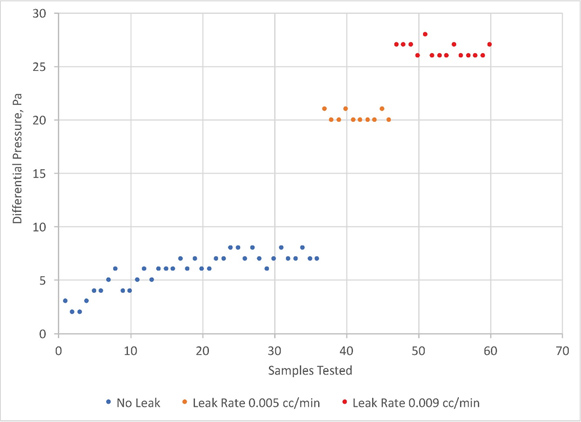
Some vacuum decay systems on the market are extremely sensitive and are capable of detecting leaks as minute as 1 µm, which effectively means a 10 µm, or even a 3-5 µm, detection is done very comfortably and reliably.9 Parenteral applications require this level of sensitivity, with the maximum allowable leakage limit (MALL) being at the single digit micron level to eliminate any risk to the patient.
But here’s a caveat: Most future applications in pharma will involve large molecules and biologics, and according to Stauffer, vacuum decay is not a reliable method for that purpose. “The reason is that these large molecules tend to clog up tiny defects and render them undetectable by vacuum-based methods,” he notes. “Also, when the product is very viscous, vacuum decay doesn’t function as the best test method and we don’t recommend it.”
For these cases, HVLD functions as an effective solution. Over the last 10 years, there have been some impressive developments in HVLD systems, and MicroCurrent HVLD (HVLDmc) is an example. In this method, a combination of AC/DC voltage is used to apply a much lower voltage to the inspection (less than 5% of the voltage applied in comparable conventional HVLD solutions).10 Not only is this less damaging to the product, but it also reduces the production of ozone during operation. “Ozone in the headspace of a container can be detrimental to the product and can be unsafe in the operating environment because it affects respiratory health,” explains Stauffer.
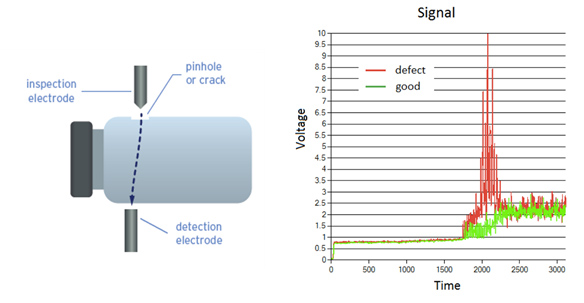
MicroCurrent HVLD also comes with the added advantage of being able to inspect highly viscous liquids, low-conductivity liquids, and all types of plastic container formats (plastic vials, syringes, etc.). For instance, water-for-injection (WFI) is extremely low in conductivity and thus poses a challenge for conventional HVLD. With MicroCurrent HVLD, the range of liquid conductivity that can be tested is very large, allowing accurate testing of WFI and other liquids even at the single micron level.
Future outlook for CCI testing
Stauffer believes that globally there is a shift toward improving both the frequency and the type of CCI testing that is performed, albeit with different approaches by different regulatory bodies.
“The European contingent is focusing on the overall requirements of the sterile finish operations,” he notes. “Annex 1 from the European Medicines Agency (EMA) provides broad concepts and ideas that call for more stringent inspection measures. But it's not that prescriptive in terms of what should be done and how it should be done.”
Comparatively, in the United States, the FDA and USP are far more prescriptive, outlining very specific methodologies for conducting CCI testing. Either way, both sides seem to be moving the needle in the direction of higher-quality, more quantitative testing.
“There are some organizations that are pushing back on these regulatory measures,” admits Stauffer. “Some organizations don’t like to move away from traditional methods, but experts and regulators both agree that it’s time we adapt methods leading to a more patient-oriented quality culture.”
References:
- Blouet, E. Parenteral Preparations, Challenges in Formulations. Pharmaceutical Outsourcing (2016).
- Booth, C. Understanding Container Closure Integrity Testing. American Pharmaceutical Review (2016).
- Wolf, H. et. al. Vacuum Decay Container/Closure Integrity Testing Technology. Part 2: Comparison to Dye Ingress Tests. PDA Journal of Pharmaceutical Science and Technology (2009), 63(5): 489–498.
- Stauffer, O., Mullan, J. Shifts in Container Closure Integrity Test Methods. Pharmaceutical Engineering (2019).
- Wolf, H. et al. Vacuum Decay Container/Closure Integrity Testing Technology. Part 1. ASTM F2338-09 Precision and Bias Studies. PDA Journal of Pharmaceutical Science and Technology (2009), 63, 472-488.
- Damgaard, R. et al. High-Voltage Leak Detection of a Parenteral Proteinaceous Solution Product Packaged in Form-Fill-Seal Plastic Laminate Bags. Part 1. Method Development and Validation. PDA Journal of Pharmaceutical Science and Technology (2013), 67, 634-651.
- Ewan, S. et al. Dye Ingress Methods for Container–Closure Integrity Testing: An Industry Position Paper. BioProcess International (2018).
- FDA recognized consensus standard ASTM F2338 – 09: Standard Test Method for Nondestructive Detection of Leaks in Packages by Vacuum Decay Method (2013).
- Transformative Shifts in Vacuum Decay Leak Testing. PTI USA (2018).
- Stauffer, O. A Paradigm Shift In Container Closure Integrity Of Parenteral Drugs. Med Device Online (2017).
- Stauffer, O. Viscous Product No Match for New CCI Tech. PDA Letter (2018), 59(7).
