Regulatory Information Management Software For Pharmaceutical Manufacturing
RImanager centralizes regulatory and product data, integrates with production, and streamlines planning, tracking, and approvals to bring new drugs to market faster and reliably.
You want to bring new drugs to the market faster and more reliably? We have the solution for you!
Körber’s RImanager is a centralized, master data-driven regulatory information management solution that allows you to easily organize your product portfolio in line with regulatory requirements.
Our software solution lets you manage your product master data as well as plan and track regulatory data, activities, processes, requirements, and approvals – across all departments and worldwide. In short, RImanager simplifies the time-consuming approval process for a new drug.
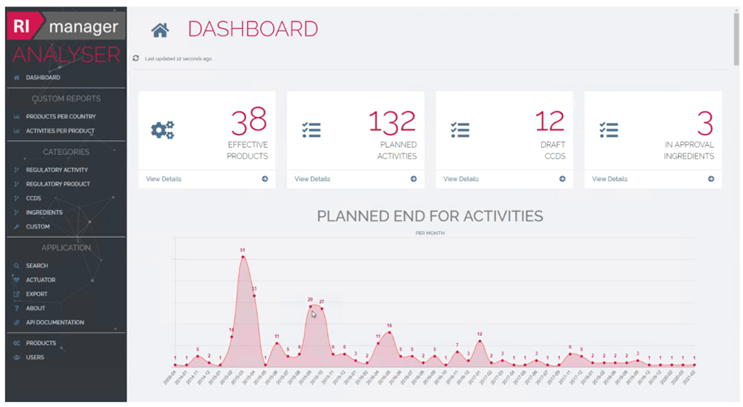
How the RImanager works: Interlinking regulatory processes with production
RImanager manages all of your products’ master data. Regulatory activities can be defined in order to save and track information about every product, including associated approval data.
As the RIM solution is built on the Werum PAS-X MES platform, it can be fully integrated into your production processes, when PAS-X MES is deployed. This ensures that all labelling, formulas, and drug safety information is in a single place. Your users can thus access the information directly and use key management and reporting functions.
The functions at a glance:
- Product master data management
- Combination of regulatory affairs and state-of-the-art technology
- Development and service provided by the leading suppliers of Manufacturing Execution Systems (MES) and regulatory affairs software
- Access to all current and archived product data via a single user interface
- Simplified planning of regulatory activities
- Creation of a single source of information for regulatory product data
How the RImanager supports you: Managing regulatory projects efficiently
In addition to regulatory data, RImanager includes powerful project management tools that help you structure and steer higher-level regulatory processes.
Integrated task management tools enable you to assign specific activities to individual departments or teams. Direct links between tasks and production sites, pharmaceutical products, formulas, and approval documents allow a fast and targeted access to related information.
For very complex processes, a workflow function can be used to assign specific tasks to different users. The fully audited workflows can also be used to manage review and approval processes. E-mail notifications ensure that users are informed in real time about upcoming tasks and deadlines.
RImanager also contains a version control feature that allows to track the status of every document within the system. For example, you can work on a new draft without changing the published version.
The services at a glance:
- Assignment of tasks and responsibilities to individual team members
- Easy creation of management reports
- Fast and simple creation of product and approval documentation
- Supports EU and US regulatory procedures
- E-mail notifications compliant with FDA CFR 21, Part 11
- Powerful project management tool for planning and tracking regulatory activities, tasks, and resources
- Provision of structured data that supports IDMP-compliant exchange (Identification of Medicinal Products) with the authorities
Your benefits
- Comprehensive control: With RImanager, you have all your processes in view at all times
- Simplified processes: New drugs can be brought to market faster and more reliably
- Effortless integration: RImanager builds on the Werum PAS-X MES platform and can be fully integrated into existing processes, when PAS-X MES is deployed
