Single-Use Flexible Containment Systems For Charging And Offloading Pharmaceutical Powder Transfer Operations

While pharmaceutical and biopharmaceutical manufacturers can choose from a broad array of containment systems, not all are suited to processes where high-level containment is critical. For OEB Level 5 containment, or for processes where personnel safety and cross-contamination are key concerns, only the highest-performing containment systems make sense. Current industry trends are moving away from multiuse stainless-steel components that require time-consuming cleaning validations.
EVALUATING ALTERNATIVES DOVERPAC — PROVEN FLEXIBLE CONTAINMENT
ILC Dover pioneered the development of flexible containment systems in 1997 and has continued to improve and refine its systems ever since. The DoverPac system was designed in collaboration with Eli Lilly to provide high levels of reliable containment during charging and offloading operations.
The system consists of a tough fabric outer restraint and a durable film liner, as well as carefully crafted O-rings, clamps, sampling ports, crimps and valves, and every component of the DoverPac system has been designed to contribute to its reliable performance and high-level containment properties. Only when all these components consistently operate as designed will the overall system provide reliable OEB Level 5 containment.
UNPROVEN NEWER SINGLE-USE VALVE SYSTEMS
In recent years, a number of single-use transfer systems have been introduced. And for some processes, where containment performance is not critical or demanding, they may perform adequately. However, several manufacturers of those systems offer no third-party performance test data to prove their performance. In fact, to achieve satisfactory containment when these systems are used as a primary isolator valve, a secondary containment scheme (an enveloping flexible isolator or PPE gear worn by operator personnel) must be employed. This adds a level of complexity and cost that makes such a system unwieldy, even if it can be designed to provide acceptable performance.
SUPERIOR OEB LEVEL 5 CONTAINMENT
SPECIFY WITH CONFIDENCE AND ELIMINATE THE NEED FOR SECONDARY CONTAINMENT
DoverPac systems’ performance is well-established by third-party testing, and you can specify with confidence, knowing the performance quoted will be the performance achieved in the plant. No additional enveloping containment or PPE equipment will be required, even to meet OEB Level 5 criteria.
ENJOY THE SECURITY OF DWELL-TESTED INFLATION
Within the walls of the ILC Dover manufacturing facility in Frederica, Delaware, hangs a sealed DoverPac film isolator, inflated in 2007. More than 10 years later, it remains inflated. No additional air has been added, and no extraordinary steps have been taken to preserve its internal pressure or control the ambient environment in that section of the plant.
Every DoverPac single-use container is visually inspected and dwell (pressure) tested to make sure it will operate as expected once it is placed into service. This enhanced level of testing is unique to ILC Dover.
DESIGN WITH THE FLEXIBILITY OF LARGE VOLUME AND HIGH-PERFORMANCE CONTAINMENT (<1μg/M3)
DoverPac systems are the only flexible, single-use containment vessels that offer capacities that range from 20 L to not just 150 L, but all the way to 2,000 L. So you can scale your operations from the lab to the pilot plant to final full-scale production, without changing containment systems or basic operations.
Further, each DoverPac containment vessel includes an integral sleeve that can be used for safe product sampling for QC analysis. The sleeve is designed to be completely sealed using our reliable CrimpLoc™ system.
PROVEN PERFORMANCE
To measure and prove their performance in critical containment applications, ILC’s DoverPac systems have been thoroughly tested for containment performance by third-party laboratories. Validation and testing were conducted using methodology established by the International Society for Pharmaceutical Engineering’s (ISPE’s) Standardized Measurement of Equipment Particulate Airborne Concentration (SMEPAC) guide, titled “Assessing the Particulate Containment Performance of Pharmaceutical Equipment.” These tests have shown that all components of DoverPac systems consistently meet the highest performance standards, maintaining containment performance levels ranging from 1μg/m3 to 4 ng/m3.
Note: All airborne-contaminant concentrations in the accompanying figures are expressed in micrograms of the surrogate test compound per cubic meter of air (μg/m3). In other words, the full quantity of airborne powder was captured in the measurements. In typical pharmaceutical applications, however, the concentration of API would be a very small fraction of the powder being processed, so the figures in the tables should represent very conservative performance expectations.
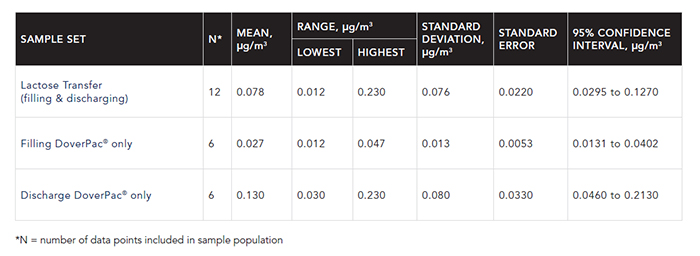
FIGURE 1: INDEPENDENT CONTAINMENT PERFORMANCE ASSESSMENT FOR DOVERPAC DISPENSING SYSTEM
Analyte: Lactose
Sample medium: 25 mm polytetrafluoroethylene filter (1.0 μm pore size) in two-piece, conductive cassette Measurement type: Personal sample breathing zone (PSBZ)
Process:
- Fill DoverPac system from vessel, crimp and seal.
- Remove crimp and discharge DoverPac system contents to vessel.
ADVANTAGES OF ILC’S DOVERPAC SYSTEMS
Unlike other films on the market, our film was designed solely for use with powders. This means it contains no additives that can discolor or contaminate your product. Furthermore, our low-residual, antistatic film ensures that all of the product gets where it’s supposed to be and doesn’t get thrown away inside the bag.
DoverPac single-use containment systems and continuous liners, on the other hand, have been designed from the ground up to suit modern powder applications. We understand this field and have listened closely to our customers to make sure our films fit the process and contribute to its efficient operation.
CRITICAL QUALITY ATTRIBUTES FOR ALL OF OUR FILMS
- No slip agents
- High film strength
- High seal strength
- Minimal film elongation
- High flex-fatigue resistance
- Abrasion- and puncture-resistant
- Gamma irradiation compatibility
While some suppliers suggest that a base film without integral static dissipative additives can be used with nothing more than an exterior surface treatment, this is simply incorrect. Coated films will not meet the intent of pharmacopoeia testing, since test monographs require that the composite cross-section of the film be tested. This is done to ensure that, in the event a piece of film should contact the drug product, the film will not introduce a potential hazard to the patient.
Because of that, static dissipative properties in ILC Dover ArmorFlex® films are achieved by the thorough blending of the antistatic compound into the base polymer during manufacturing. This creates a homogeneous conductive network yielding consistent antistatic properties throughout the film.
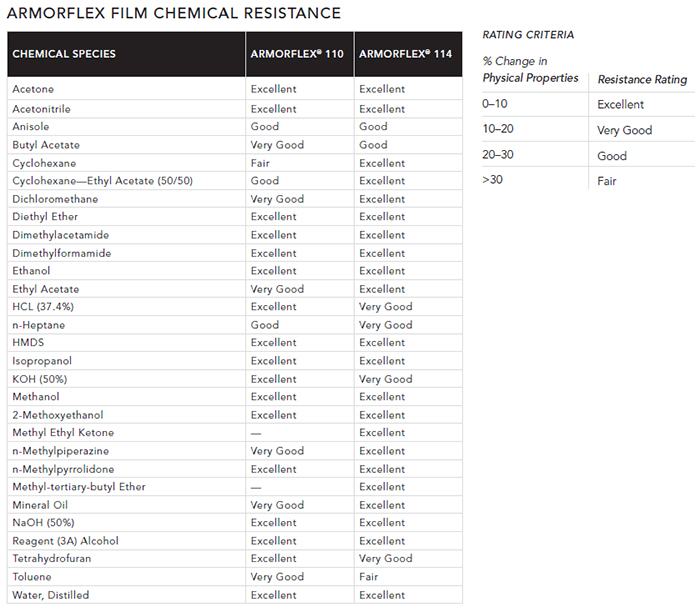
ARMORFLEX FILMS PROVIDE A FULL RANGE OF PERFORMANCE AND CHEMICAL RESISTANCE
ArmorFlex is ILC Dover’s proprietary family of films used exclusively in the DoverPac family of high-efficiency flexible containment products. DoverPac containment products can be manufactured from one of two high-performance polymer films. Each offers a specific set of characteristics that make that film ideal for certain applications. The choices include ArmorFlex 110 or ArmorFlex 114 films. Their characteristics are summarized below.
ARMORFLEX 110
A robust film developed to respond to evolving regulations, ArmorFlex 110 film delivers permanent static dissipative properties while meeting FDA and 2002/72/EC requirements for food contact. It also complies with FDA 21 CFR and passes USP <661> Physicochemical Tests for Plastics, as well as USP <88> Class VI (7-day implant), <87> Biological Reactivity (in vitro) and Chilworth incendivity tests. It offers a five-year shelf life, and its permanent antistat maintains the integrity of our users’ products.
ARMORFLEX 114
Our newest antistatic film, ArmorFlex 114, represents the most advanced film we have developed to date. It passes Chilworth incendivity tests has strong chemical resistance characteristics; complies with FDA 21 CFR, 2002/72/EC and amendments; meets EP 3.1.3 test parameters; and passes USP <661> Physicochemical Tests for Plastics, USP <88> Class IV (7-day implant) and USP <87> Biological Reactivity (in vitro). It also offers a five-year shelf life, and a DMF has been filed with the FDA.
THE CRIMPLOC SEALING SYSTEM IS KEY TO CONTAINMENT PERFORMANCE
The ILC Dover CrimpLoc sealing system delivers unrivaled high-containment performance in a flexible single-use system. The CrimpLoc system is reliable — much more so than twist-tape-cut procedures or the use of zip ties, both of which are much more dependent on operator technique. No matter which ArmorFlex film is chosen, ILC’s CrimpLoc system will ensure the highest containment performance.
The CrimpLoc seal provides secure closure and easy, one-piece crimp removal without any breaking or risk of plastic shards dropping into the product. The containment performance of our CrimpLoc system has been tested and proven to provide API and HPAPI powder transfers at containment performance targets (CPTs) ranging from cGMP to nanogram levels.
COMPARING COSTS
DoverPac single-use containment systems have very low capital costs compared to other technologies, as well as reasonable cost of consumables, which combine to provide a very economical total life-cycle cost.
Systems using stainless steel split butterfly valves, on the other hand, are subject to high front-end cost and complex cleaning/verification. The cost and complexity of these valves increases significantly based on the OEB level that is required due to adding vacuum purging or electronic valve sequencing, and status reporting.
UNPROVEN SINGLE-USE TRANSFER VALVE SYSTEMS
Several new single-use valves are being tried in the market. Not all of them have undergone robust SMEPAC containment testing. In addition, most are only available in a 4-inch valve size and do not offer true full-port flow. The valve sizes limit the charge bag volumes to less than 100 L. In contrast, DoverPac transfer systems offer 12-inch or 8-inch transfer sleeves, and are available in volumes up to 2,000 L.
Single-use slide-gate valves have shown unreliable performance for some pharmaceutical industry users. Improvements to the design have been made, but consistent reliability has yet to be proven through field performance.
Single-use cone valves have some early usage. While the polymer cone that is integrated into the charge bag is relatively inexpensive, the active portion of the valve can cost between $50,000 and $100,000, depending on the level of sophistication.
Single-use split butterfly valves are relatively new to the market and are just starting to be used. Field reliability is still being established, and containment performance testing is not yet available.
Disposable flat-pinch valves may work the first time, but repeated undocking can lead to powder accumulation between the upper and lower valves. This style of connection also requires special equipment to position and hold the connectors, and then force the valves open.
THE COMFORT OF DOVERPAC SYSTEMS
By contrast, DoverPac single-use systems with CrimpLoc technology have established their reliability through years of operation in all kinds of pharmaceutical applications, with full SMEPAC-tested containment performance data to back up their reliability.
With product and worker safety on the line, don’t take a chance with unproven technology. Demand containment performance test results. The best solutions are reliable, simple and time tested.
ABOUT US
Since 1947, ILC Dover has built a global reputation for out-of-the-box thinking that makes the seemingly impossible possible. Our engineered solutions solve our customers’ most complex challenges through the creative and efficient application of flexible materials often integrated with advanced equipment
and hardware.
We look beyond the boundaries of convention to help customers see what could be, and discover the extraordinary possibilities within everyday things. We are a diverse company serving many markets. We are dreamers, engineers, scientists and pragmatists — all dedicated to outperforming tradition to better mankind.
We apply our vast knowledge of materials, soft goods, film-based solutions and design to move the world forward, from advancing spacesuits for astronauts to developing solutions for NASA Mars missions to engineering lighter-than-air vehicles here on Earth. We continue to pioneer the use of flexible containment solutions to support advanced pharmaceutical and biopharmaceutical manufacturing, and we’re revolutionizing the packaging and extraction of bulk liquids to enhance customer profitability
and sustainability.
Additionally, we create quick-deploy systems that protect cities and critical infrastructure from floods, and design and manufacture advanced respirators to protect against a range of chemical and biological threats.
Every day, everything we do brings new solutions to light. Are you ready to take your vision beyond boundaries? Let’s talk.
