ABOUT MASTERCONTROL
MasterControl Inc. is a leading provider of cloud-based Quality Management System (QMS) and Manufacturing Execution System (MES) for life sciences and other regulated industries. Our mission is the same as that of our customers – to bring life-changing products to more people sooner. The MasterControl Platform helps organizations digitize, automate and connect critical processes across the regulated product development life cycle.
QUALITY MANUFACTURING SOFTWARE E-BOOKS
-
This guide will help you understand the fundamental components of Quality Management System (QMS) software and how QMS solutions are evolving to keep pace with business dynamics.
-
How adopting a data-driven and technology-supported approach to quality and product life cycle management can enable pharma and biotech organizations to not just survive, but thrive.
-
The digital documentation layer of quality and compliance processes is still critical and will never go away, but advanced technologies are helping life sciences organizations unlock the data side of the equation.
-
This ebook explores the current landscape of the medical device industry and the forces taking shape today that will drive growth into 2030 and beyond. From new technology to shifting patients’ experiences, we’ll explore how a wide range of issues are affecting product development and innovation, as well as the impact of digitization and data when it comes to device regulations and operations.
-
Read how a smarter, faster and smaller approach to eliminating paper processes could provide the evidence people seek to pursue a digital transformation.
-
MasterControl is the foundation for connected quality data and complete product quality. It unifies applications, data, and documentation across your entire product development life cycle, from concept to commercialization. Go beyond proactive quality management and unleash the intelligence and insights concealed in unstructured data. Discover how the MasterControl Platform can make your vision of truly connected quality data a reality.
CONTACT INFORMATION
MasterControl, Inc.
6350 South 3000 East
Salt Lake City, UT 84121
UNITED STATES
Phone: Sales 1.888.873.5164
MANUFACTURING SOFTWARE E-BOOKS
-
Gain an understanding of how leading pharmaceutical manufacturers are modernizing their operations, how to stay current with industry trends, and ensure long-term relevance in the value chain.
-
With so many manufacturing software products on the market today, evaluating and choosing the right one for your organization can be overwhelming. It doesn't have to be if you know the essential capabilities you need.
-
Explore the state of digital transformation in manufacturing and learn how you can extend your digital edge.
BROCHURES
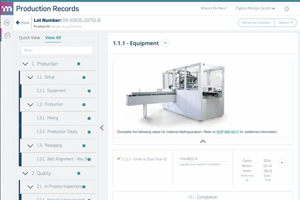
- Digital Production Records For Modern Manufacturing
- MasterControl Change Control™
- MasterControl Manufacturing Excellence™ Systems Integrations
- MasterControl Recipe™
- MasterControl Electronic Batch Records™ (EBR)
- Digital Production Records For Modern Manufacturing
- The Next Generation Of The Number One Enterprise Quality Management System (QMS)
- Product Lifecycle Excellence Platform
- Ensuring Compliance, Improving Efficiency, And Accelerating Time To Market With QMS Software
VIDEOS
- Accelerate Innovation With Compliant Software Solutions
- Join Us At Summit CXO
- A QMS That Helps You Transform Compliance Into A Competitive Advantage
- A Modern Pharma MES Solution Offers The Best Of Old And New
- EpiBone: Personalized Medicine. Built On Quality.
- MasterControl EBR & Advantages Of Digitizing Batch Records
- Life Sciences Quality And Compliance Software Product Demonstration
FEATURED ARTICLES
-
Explore the manufacturing trends shaping 2026, from AI in validated environments to connected systems and digital workforce readiness that are building more agile, resilient, and compliant operations.
-
Explore how cell and gene therapy manufacturers are scaling from batch‑of‑one to commercial supply while staying compliant, digital, and efficient, as well as strategies to strengthen quality.
-
Learn how digital maturity and compliance excellence are reshaping CDMO competitiveness, as well as strategies that strengthen performance and accelerate tech transfers.
-
Discover how the updated ICH E6(R3) guideline supports the speed and complexity of CGT manufacturing, enabling flexible, risk‑based decisions and quality systems built to scale therapies safely.
-
Explore how digital systems strengthen CGT manufacturing by improving traceability and managing batch‑of‑one, as well as essential steps toward scalable, integrated operations.
-
Learn how outcome-based automation helps life sciences manufacturers reduce risk, improve performance, and drive measurable results—by aligning tools with business goals.
-
Life sciences compliance is complex and ever-changing. Discover how modern solutions can streamline processes, reduce friction, and keep your organization ahead of regulatory demands.
-
BioBridge Global faced challenges with paper-heavy processes that risked errors and delays. Discover how digital solutions streamlined their operations and improved regulatory efficiency.
-
Discover how evolving global regulations are reshaping CDMO strategy. and how digital infrastructure helps manufacturers stay audit-ready, build sponsor trust, and turn compliance into an advantage.
-
Explore how modern manufacturing software is transforming operations with AI-driven insights to empower CDMOs and CMOs to boost efficiency, agility, and client satisfaction in a competitive landscape.
-
Cell and gene therapy has evolved from a niche concept to a booming sector in just five years, with over 1,300 companies and growing momentum in innovation, investment, and regulatory approvals.
-
Life sciences manufacturers face growing pressure to adopt AI while staying compliant. Discover five key areas to evaluate AI tools and how to simplify adoption with regulatory assurance.
-
Cell and gene therapies are revolutionizing medicine, but outdated manufacturing methods are slowing progress. Discover how modernizing CGT production can unlock the full potential of life-changing treatments.
-
Innovators in life sciences manufacturing drive life-saving breakthroughs. Amid strict regulations and complex challenges, learn how your dedication ensures quality and pushes the boundaries of what’s possible in public health.
-
Life sciences manufacturing is evolving rapidly. Discover how digital transformation is revolutionizing production processes to enable personalized medicine and improve patient outcomes across the healthcare industry.
-
Companies still relying on paper-based manufacturing are falling behind. Examine how digital transformation boosts efficiency and reduces errors in this essential shift toward smarter operations.
-
Discover how PCI Pharma Services embraced digital transformation and gain expert insight on overcoming challenges, driving innovation, and maintaining core values in a regulated industry.
-
Review contract manufacturing's evolution, driven by technological advancements and regulatory changes, and how CMOs and CDMOs are navigating opportunities and challenges.
-
Regulated industries face rising demand, tech breakthroughs, and regulatory harmonization. Discover how contract manufacturers must leverage AI and cloud solutions to stay agile and compliant in the future.
-
Discover the advantages of outsourcing equipment calibration to ISO 17025 accredited suppliers, including cost efficiency and regulatory assurance, in our detailed analysis. Read more to transform your calibration processes.
-
Life sciences startups must integrate compliance early to ensure regulatory approval, and trust. Prioritizing compliance streamlines processes, and enables long-term success in this highly regulated industry.
-
Artificial Intelligence (AI) is becoming more prevalent today and will play a crucial role in the future. Discover the latest AI trends revolutionizing quality management systems in life sciences.
-
Did you know that FDA recalls have increased 115% since 2018? Learn more on how to avoid common recall mistakes and effective recovery strategies for your company today.
-
Explore the need for digital quality management systems to ensure compliance as life science companies face rising FDA recalls.
-
Optimizing pharmaceutical product release requires effective planning, specification development, electronic systems, and collaboration with CMOs, ensuring compliance, efficiency, and quality in the drug manufacturing process.
-
Discover how life science manufacturers are supported in digital transformation, offering solutions for automation, data integration, and compliance to optimize operations.
-
IQ, OQ, and PQ validations ensure equipment reliability, compliance, and quality in medical device and pharmaceutical manufacturing, meeting regulatory standards.
-
Discover the transformative power of connected quality and manufacturing systems, offering strategies for digital transformation, enhanced efficiency, compliance, and real-world success in life sciences.
-
Pharma product release ensures drug quality, safety, and regulatory compliance by evaluating manufacturing and quality processes. It’s a critical, comprehensive process essential for FDA approval and patient safety.
-
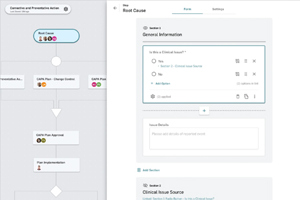
Discover why an optimized CAPA process is crucial for life sciences companies to enhance quality, ensure regulatory compliance, prevent product recalls, and maintain market competitiveness.
-
An outdated QMS limits growth. Explore how upgrading to an advanced, configurable system like MasterControl can ensure scalability, automation and innovation.
-
When selecting a QMS, it may feel like the most challenging step is making the purchase. This is not necessarily the case. Read more about the challenges of implementing an effective QMS.
-
Investigate the factors contributing to FDA recalls, the effects on both consumers and businesses, and how integrated digital solutions can play a crucial role in preventing such incidents.
-
Watch to gain expert insights on utilizing a closed-loop, single-platform approach and explore practical examples and data showcasing the advantages of integrating your QMS and MES.
-
Embrace digital transformation in pharma manufacturing with advanced document control and quality management systems to enhance compliance, streamline processes, and accelerate time to market.
-
In a rapidly evolving industry, see why adopting digital solutions is essential for achieving operational excellence and staying competitive.
-
Discover why manufacturers must adopt more consumer-centric recall strategies to maintain trust and safeguard their market position.
-
Transform your pharmaceutical manufacturing processes and enhance compliance by implementing a modern pharma manufacturing execution system (MES).
-
Avoid FDA recalls and safeguard your life sciences business. Learn how QMS and EBR/eDHR solutions can prevent costly product recalls and ensure compliance with FDA regulations.
-
With advanced features and better visibility into operations, uncover how MasterControl helps organizations navigate the complexities of the life sciences industry.
-
Take the next step in enhancing your pharmaceutical manufacturing processes by investing in a robust quality management system (QMS) designed for ICH compliance and operational excellence.
-
FDA product recalls pose significant challenges for life sciences companies. Avoid recalls with these actionable steps to mitigate risk, ensure compliance, and safeguard your organization's reputation.
-
Explore results from a study aiming to understand why numerous companies continue to rely on manual, paper-based systems, and gain insight into how your competitors are gaining a competitive advantage.
-
Not every quality management system (QMS) has the same capabilities and connectivity. Review several features to take into consideration when selecting a QMS system for your organization.
-
Generative AI is reaching a critical juncture in the life sciences industry. Learn how it is changing the game for GxP Compliance and enabling organizations to improve operational efficiency.
-
Explore the importance of supplier quality management, relevant standards, regulatory audits, best practices, and the overall significance of this practice within the life sciences sector.
-
For risk analysis to be successful, it must be carried out comprehensively from beginning to end. A holistic, integrated approach is essential, along with the adoption of appropriate tools.
-
Learn about three key regulatory differences between the United States and the European Union that can significantly impact the success of your market entry.
-
What do legacy manufacturing execution systems (MESs) lack? What can a modern pharma MES solution do? Explore the advantages of using a modern MES to evaluate the efficiency of your production line.
-
Learn about the advantages of embedding quality throughout design and manufacturing, explore five practical elements of QbD in pharmaceuticals, and discover how a QMS can support QbD.