ABOUT PARTICLE MEASURING SYSTEMS
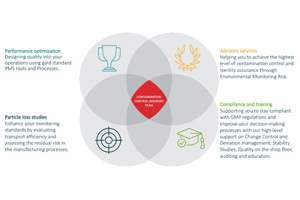
Particle Measuring Systems (PMS) provides Pharmaceutical aseptic and controlled manufacturing customers with the superior knowledge, technology, quality, and service you need to successfully meet global regulatory requirements and manufacture your product to the highest quality possible. Partnering with our customers, we provide a complete contamination monitoring solution. Starting with our Advisory Services team to assess risk, then collecting data with our world-class viable and non viable particle counters, through to compliant data management, analysis and reporting. We have the expertise to help you meet global regulatory requirements and get your product to market. As one of the world’s leading clean room contamination control experts and complete solutions provider, we offer fully integrated and robust monitoring solutions including:
- Particle Counters and Microbial Samplers, portable/mobile and remote/continuous
- Environmental and Facility Monitoring Systems (FMS)
- Data collection and fully compliant data management
Additionally, we offer a wide variety of expert Advisory Services including:
- Sampling locations
- Risk Assessments – avoiding 483’s
- Thermal Validations
- Crisis Handling – 483 management and response
Founded over 50 years ago, PMS is a trusted thought-leader in the Pharmaceutical clean manufacturing and other Life Science industries. Our team provides multiple webinars, papers, and training throughout the year to support professionals and cleanroom manufacturers with reliable guidance on the application of and compliance to global regulations.
CONTACT INFORMATION
Particle Measuring Systems
5475 Airport Blvd.
Boulder, CO 80301
UNITED STATES
Phone: 303-443-7100
Contact: Nina Morton
FEATURED ARTICLES
-
Transition from periodic sampling to continuous monitoring. Learn to establish risk-based alert limits and use N:M trending to maintain a true state of control in critical cleanroom zones.
-
Maintaining high-quality water for injection is essential for pharmaceutical safety. Discover how strategic design, proactive maintenance, and risk-based monitoring ensure reliable results.
-
Discover how to adapt your environmental monitoring framework to meet the distinct risk-management and regulatory needs of ATMP facility production.
-
Accurate cleanroom particle monitoring requires precise isokinetic sampling. Understand how to maintain sampling accuracy and meet regulatory airflow requirements for your facility.
-
Transitioning from periodic testing to continuous monitoring is essential for modern aseptic safety. Learn how real-time particle tracking mitigates risk and ensures compliance.
-
Implement robust contamination control strategies that align with current regulatory requirements. Optimize your environmental monitoring framework through precise design and data-driven analysis.
-
Maintain rigorous pharmaceutical cleanroom standards through strategic environmental monitoring. Explore essential guidelines for particle detection and effective data analysis to ensure compliance.
-
Avoid common sampling errors and ensure cleanroom compliance. Master correct probe placement and tubing configurations to align with the latest industry standards and improve data accuracy.
-
Creating a robust contamination control strategy is essential for pharmaceutical manufacturing. Learn to align risk management and technical controls to ensure lasting regulatory compliance.
-
Microbial survival during gas decompression is a proven risk. Learn why point-of-use monitoring is essential for Annex 1 compliance and how to ensure process gas purity in sterile environments.
WEBINARS
-
Maintaining the integrity of Water for Injection (WFI) systems is a non-negotiable priority in pharmaceutical manufacturing. Watch this webinar to refine your contamination control strategies.
-
Master the complexities of aseptic gowning and Annex 1 compliance. Explore risk-based strategies to mitigate human-source contamination and strengthen your facility's sterility assurance.
-
Modern aseptic monitoring requires a shift from periodic checks to risk-based strategies. Learn how to integrate particle and environmental data to meet Annex 1 standards and ensure patient safety.
-
Explore how tubing configuration impacts particle recovery and compliance under Annex 1, using evidence-based guidance, risk assessment tools, and practical strategies to strengthen contamination control.
-
Learn how automated environmental monitoring strengthens contamination control strategies, reduces risk, and supports intervention planning in aseptic filling lines.
-
Discover how modern strategies like the Contamination Control Strategy are transforming environmental monitoring by exposing hidden risks, enhancing data quality, and unlocking deeper insights through innovative microbial methods.
-
Watch how a single-use microbial impactor revolutionizes contamination control in robotic filling lines to enhance sterility, efficiency, and compliance in fully automated manufacturing environments.
-
Modern environmental monitoring systems are vital for pharmaceutical manufacturing. Explore the benefits of a tool for data collection, analysis, and processing that ensures compliance and adaptability.
-
Annex 1 mandates a risk assessment to identify optimal sample point locations. Explore factors in risk assessment, equipment selection, and system design to maximize data value.
-
There are several factors associated with active environmental microbial monitoring. Watch to learn how the EU GMP Annex 1 compliance focuses on microbial monitoring in cleanrooms.
-
Industry expert Mark Hallworth guides viewers through the latest ISO document (ISO 14644), exploring the reasons behind the decisions made by the team during its development.
-
Airflow visualization techniques like Smoke Studies and CFD analyses are crucial for contamination control, aiding cleanroom qualification, environmental monitoring, and optimizing contamination control strategies under Annex 1.
-
Discover how a Quality by Design approach can streamline the development of new sterile product filling lines, featuring a real-life example illustrating its effectiveness.
-
Discover the importance of compressed gas monitoring in ensuring product quality and compliance with EU GMP standards.
-
Review the standards and regulations promoting a cohesive environmental program, and explore manual compounding in laminar flow hoods, through to filling lines, in both open and closed systems.
-
Gain insight into the standard for microbiological monitoring and control, ISO/EN 17141, and how it harmonizes with ISO 14698 parts one and two.
-
Review the latest revisions to the USP<1788>, which has expanded to support a holistic view of finished product contaminants, how they should be investigated, and added flow imaging technologies.
-
In this webinar we review how to be compliant with the most recent particle counter calibration requirements, particle counter terminology, and how a particle counter works in theory.
-
Modern pharmaceutical manufacturers are already generating and managing big data from environmental monitoring; the next step is to create actionable insights and ensure 21CFR11 compliance. This webinar discusses data collection, 21CFR11 data management and the generation of actionable insights.
-
The New Annex 1 has some critical changes that are important to understand and start applying. While the drafts cover many areas of environmental monitoring in-depth, this webinar by Particle Measuring Systems (PMS) experts focuses on the Viable/Microbial Air Monitoring aspects.
-
Sometimes we focus on data while forgetting how poor practices, bad decisions, incorrect definitions, and lack of evaluations effect the outcome. In this webinar, we analyze how to go from “data” to “data quality” to improve your overall contamination control strategy.
-
Settle plates are a non-validatable method as it is simply based on the physical principle of the downfall of a particle on a surface. We discuss the using active air sampling versus passive air settle plate monitoring in routine environmental monitoring.
-
Defining a Contamination Control Strategy (CCS) is unique for each process and requires an intimate understanding of both the process and how to implement an effective strategy.
PODCASTS
- S2E4: Marc And Mark: Particle Monitoring In Heat Tunnels - Crucial Insights
- Airborne Particle Sampling Techniques
- Manufacturing Contamination Control Strategy And Risk Assessment
- Environmental Risk Assessment: PQ Systems And Tools
- Facility Monitoring Systems (FMS) vs. Building Management Systems (BMS)
- Tackling Viable And Non Viable Isolator Manufacturing
- Continuous Viable Air Monitoring For EU GMP Annex 1
- Risk Assessment When Setting Microbiological Monitoring Conditions
- Working Towards NetZero Emissions In Pharma Manufacturing